Two Carnot engines $A$ and $B$ are operated in series. The first one, $A,$ receives heat at $T_1(= 600\,K)$ and rejects to a reservoir at temperature $T_2.$ The second engine $B$ receives heat rejected by the first engine and, in turns, rejects to a heat reservoir at $T_3 (=400\,K).$ Calculate the temperature $T_2$ if the work outputs of the two engines are equal ..... $K$
JEE MAIN 2019, Medium
$W_1 = W_2$
$\Rightarrow \,\,\,600-T_2\,=\,T_2\,-\,400$
$\Rightarrow T_2\,=\,500\,K$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Volume versus temperature graph of two moles of helium gas is as shown in figure. The ratio of heat absorbed and the work done by the gas in process $1-2$ isView Solution
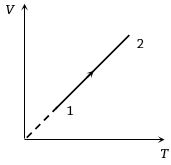
- 2An ideal heat engine exhausting heat at ${77^o}C$ is to have a $30\%$ efficiency. It must take heat at ...... $^oC$View Solution
- 3View SolutionWhich of the following is not a thermodynamics co-ordinate
- 4In a thermodynamic process pressure of a fixed mass of a gas is changed in such a manner that the gas releases $20 \,J$ of heat when $8 \,J$ of work was done on the gas. If the initial internal energy of the gas was $30 \,J$, then the final internal energy will be ........ $J$View Solution
- 5A motor-car tyre has a pressure of $2\, atm$ at $27\,^oC$. It suddenly burst's. If $\left( {\frac{{{C_p}}}{{{C_v}}}} \right) = 1.4$ for air, find the resulting temperatures (Given $4^{1/7} = 1.219$)View Solution
- 6$540$ calories of heat converts $1$ cubic centimeter of water at $100\,^oC$ into $1671$ cubic centimeter of steam at $100\,^oC$ at a pressure of one atmosphere. Then the work done against the atmospheric pressure is nearly .......... $cal$View Solution
- 7One mole of a monatomic ideal gas undergoes the cyclic process $J \rightarrow K \rightarrow L \rightarrow M \rightarrow J$, as shown in the $P - T$ diagram.View Solution
Match the quantities mentioned in $List-I$ with their values in $List-II$ and choose the correct option. [ $R$ is the gas constant]
$List-I$ $List-II$ ($P$) Work done in the complete cyclic process ($1$) $R T_0-4 \ R T_0 \ln 2$ ($Q$) Change in the internal energy of the gas in the process $JK$ ($2$) $0$ ($R$) Heat given to the gas in the process $KL$ ($3$) $3 \ R T_0$ ($S$) Change in the internal energy of the gas in the process $MJ$ ($4$) $-2 \ R T_0 \ln 2$ ($5$) $-3 \ R T_0 \ln 2$ 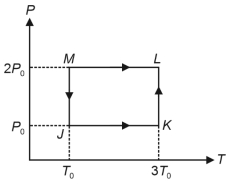
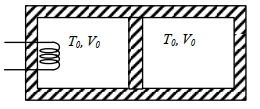
- 8A thermally insulating cylinder has a thermally insulating and frictionless movable partition in the middle, as shown in the figure below. On each side of the partition, there is one mole of an ideal gas, with specific heat at constant volume, $C_v=2 R$. Here, $R$ is the gas constant. Initially, each side has a volume $V_0$ and temperature $T_0$. The left side has an electric heater, which is turned on at very low power to transfer heat $Q$ to the gas on the left side. As a result the partition moves slowly towards the right reducing the right side volume to $V_0 / 2$. Consequently, the gas temperatures on the left and the right sides become $T_L$ and $T_R$, respectively. Ignore the changes in the temperatures of the cylinder, heater and the partition.View Solution
($1$) The value of $\frac{T_R}{T_0}$ is
$(A)$ $\sqrt{2}$ $(B)$ $\sqrt{3}$ $(C)$ $2$ $(D)$ $3$
($2$) The value of $\frac{Q}{R T_0}$ is
$(A)$ $4(2 \sqrt{2}+1)$ $(B)$ $4(2 \sqrt{2}-1)$ $(C)$ $(5 \sqrt{2}+1)$ $(D)$ $(5 \sqrt{2}-1)$
Give the answer or qution ($1$) and ($2$)
- 9Initial pressure and volume of a gas are $P$ and $V$ respectively. First it is expanded isothermally to volume $4V$ and then compressed adiabatically to volume $V$ . The final pressure of gas will be (given $\gamma = 3/2$ )View Solution
- 10In an adiabatic process, the density of a diatomic gas becomes $32$ times its initial value. The final pressure of the gas is found to be $n$ times the initial pressure. The value of $n$ isView Solution
