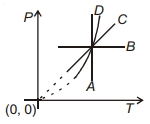
Following figure shows $P-T$ graph for four processes $A, B, C$ and $D$. Select the correct alternative.

Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An ideal gas at pressure $P$ and volume $V$ is expanded to volume$ 2V.$ Column $I$ represents the thermodynamic processes used during expansion. Column $II$ represents the work during these processes in the random order.:View Solution
Column $I$ Column $II$ $(p)$ isobaric $(x)$ $\frac{{PV(1 - {2^{1 - \gamma }})}}{{\gamma - 1}}$ $(q)$ isothermal $(y)$ $PV$ $(r)$ adiabatic (z) $PV\,\iota n\,2$ The correct matching of column $I$ and column $II$ is given by
- 2One mole of an ideal gas undergoes a cyclic process, consisting of two isochores and two isobars. Temperature at $1$ and $3$ equal to $T_1$ and $T_3$ respectively. The work done by the gas over the cycle, if the point $2$ and $4$ lie on the same isothermView Solution
- 3Heat given to a system is $35$ joules and work done by the system is $15$ joules. The change in the internal energy of the system will be ..... $J$View Solution
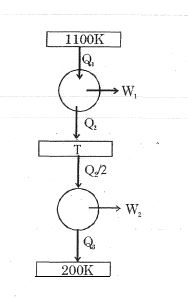
- 4Consider the given series combination of carnot cycles. If $W_1 = W_2$ then the value of $T$ is ...... $K$ (all temperatures are maintained at their respective values)View Solution
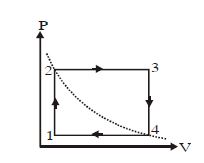
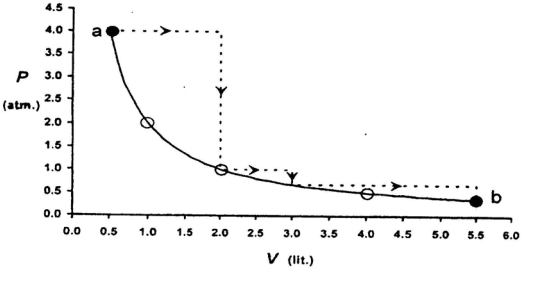
- 5One mole of an ideal gas is taken from a to $b$ along two paths denoted by the solid and the dashed lines as shown in the graph below. If the work done along the solid line path is $\mathrm{w}_{\mathrm{s}}$ and that along the dotted line path is $w_d$, then the integer closest to the ratio $w_d / w_5$ isView Solution
- 6View SolutionIn isothermic process, which statement is wrong
- 7$200\,g$ water is heated from $40\,^oC$ to $60\,^oC.$ Ignoring the slight expansion of water, the change in its internal energy is close to ...... $kJ$ (Given specific heat of water $=4184\,J/kgK$ )View Solution
- 8A Container having $1$ mole of a gas at a temperature $27°C$ has a movable piston which maintains at constant pressure in container of $1 \,atm.$ The gas is compressed until temperature becomes $127°C$. The work done is ........ $J$ ($C_P$ for gas is $7.03\, cal/mol-K)$View Solution
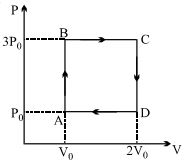
- 9A ideal monoatomic gas is carried around the cycle $ABCDA$ as shown in the fig. The efficiency of the gas cycle isView Solution
- 10A Carnot engine has efficiency $25\%$ . It operates between reservoirs of constant temperature with temperature difference of $80\,K$ . What is the temperature of low temperature reservoir ...... $^oC$View Solution