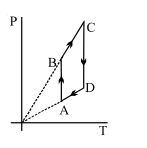
Two moles of helium gas are taken over the cycle $ABCDA$, as shown in the $P-T$ diagram. The net work done on the gas in the cycle $ABCDA$ is ...... $R$


AIEEE 2009, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionPressure versus temperature graph of an ideal gas is shown in figure
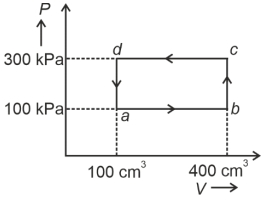
- 2A cyclic process of a thermodynamic system is taken through $a$ $b$ $c$ $d$ $a$. The work done by the gas along the path $b$ $c$ isView Solution
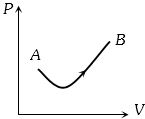
- 3View SolutionConsider a process shown in the figure. During this process the work done by the system
- 4View SolutionThe state of a thermodynamic system is represented by
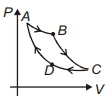
- 5Carnot cycle is plotted in $P-V$ graph. Which portion represents an isothermal expansion?View Solution
- 6An ideal gas goes through a reversible cycle $a\to b\to c\to d$ has the $V - T$ diagram shown below. Process $d\to a$ and $b\to c$ are adiabatic.... The corresponding $P - V$ diagram for the process is (all figures are schematic and not drawn to scale)View Solution
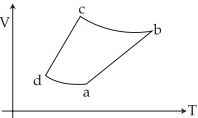
- 7View SolutionA gas is compressed adiabatically, which one of the following statement is NOT true.
- 8For an ideal heat engine, the temperature of the source is $127\,^{\circ} C$. In order to have $60\, \%$ efficiency the temperature of the sink should be $........\,{ }^{\circ} C$. (Round off to the Nearest Integer)View Solution
- 9The amount of work done in an adiabatic expansion from temperature $T$ to ${T_1}$ isView Solution
- 10An ideal gas, initially in state $\left( P _{12}, V _1, T _1\right)$ is expanded isobarically to $\left( P _{12}, V _2, T _2\right)$, then adiabatically $\left( P _{34}, V _3, T _3\right)$. It is then contracted isobarically to $\left( P _{34}, V _4, T _4\right)$ and finally adiabatically back to the initial state. The efficiency of this cycle isView Solution
