A mixture of ideal gases has $2$ moles of He, $4$ moles of oxygen and $1$ mole of ozone at absolute temperature $T$. The internal energy of mixture is ............. $RT$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Find the ratio of specific heat at constant pressure to the specific heat constant volume for $N{H_3}$View Solution
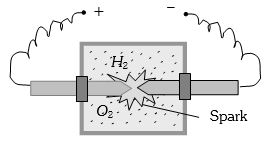
- 2A vessel contains $14 \,gm \,(7 $ moles) of hydrogen and $96\, gm$ ($9$ moles) of oxygen at $STP.$ Chemical reaction is induced by passing electric spark in the vessel till one of the gases is consumed. The temperature is brought back to it's starting value $273 K.$ The pressure in the vessel is ...... $atm$View Solution
- 3$c_P$ and $c_V$ are specific heats at constant pressure and constant volume respectively. It is observed thatView Solution
$c_P - c_V = a$ for hydrogen gas
$c_P - c_V = b$ for nitrogen gas
The correct relation between $a$ and $b$ is
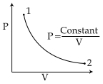
- 4View SolutionWhich one the following graphs represents the behaviour of an ideal gas
- 5The volume of a gas at $21°C$ temperature and $768 \,mm$ pressure is $1\, litre$. If the density of the gas is $1.2\, gm/litre$ at $NTP,$ then its mass will be ..... $gm$View Solution
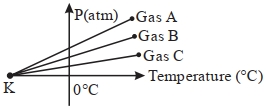
- 6For three low density gases $A, B, C$ pressure versus temperature graphs are plotted while keeping them at constant volume, as shown in the figure.View Solution
The temperature corresponding to the point ' $K$ ' is $...........\,{}^{\circ}\,C$
- 7In a vessel, the gas is at pressure $P_0$. If the mass of all the molecules is halved and their speed is doubled, then the resultant pressure will beView Solution
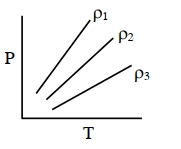
- 8$P-T$ diagram of an ideal gas having three different densities $\rho_1, \rho_2, \rho_3$ (in three different cases) is shown in the figure. Which of the following is correct :View Solution
- 9A gas at absolute temperature $300\,K$ has pressure $= 4 \times 10^{-10}\,N /m^2$ . Boltzmann constant, $k = 1.38 \times 10^{-23}\,J / K$ . The number of molecules per $cm^3$ is of the order ofView Solution
- 10For the $P-V$ diagram given for an ideal gas, out of the following which one correctly represents the $T-P$ diagram ?View Solution