$f=\frac{\Delta \mathrm{U}}{(\Delta \mathrm{Q})_{\mathrm{P}}}=\frac{(\Delta \mathrm{Q})_{\mathrm{V}}}{(\Delta \mathrm{Q})_{\mathrm{P}}}=\frac{\mu \mathrm{C}_{\mathrm{V}} \Delta \mathrm{T}}{\mu \mathrm{C}_{\mathrm{P}} \Delta \mathrm{T}}=\frac{1}{\gamma}$
For diatomic gas $\gamma=\frac{7}{5} \Rightarrow f=\frac{5}{7}$
Download our appand get started for free
Similar Questions
- 1The coefficient of performance of a Carnot refrigerator working between ${30^o}C$ and ${0^o}C$ isView Solution
- 2The equation of state for a gas is given by $PV = nRT + \alpha V$, where $n$ is the number of moles and $\alpha $ is a positive constant. The initial temperature and pressure of one mole of the gas contained in a cylinder are $T_o$ and $P_o$ respectively. The work done by the gas when its temperature doubles isobarically will beView Solution
- 3This question has Statement $1$ and Statement $2.$ Of the four choices given after the Statements, choose the one that best describes the two Statements.View Solution
Statement $1:$ In an adiabatic process, change in internal energy of a gas is equal to work done on/by the gas in the process.Statement $2 :$ The temperature of a gas remains constant in an adiabatic process.
- 4$1c{m^3}$ of water at its boiling point absorbs $540$ calories of heat to become steam with a volume of $1671c{m^3}$.If the atmospheric pressure = $1.013 \times {10^5}N/{m^2}$ and the mechanical equivalent of heat = $4.19J/calorie$, the energy spent in this process in overcoming intermolecular forces is ..... $cal$View Solution
- 5One mole of an ideal monoatomic gas undergoes the following four reversible processes:View Solution
Step $1$ It is first compressed adiabatically from volume $V_{1}$ to $1 \;m ^{3}$.
Step $2$ Then expanded isothermally to volume $10 \;m ^{3}$.
Step $3$ Then expanded adiabatically to volume $V _{3}$.
Step $4$ Then compressed isothermally to volume $V_{1}$. If the efficiency of the above cycle is $3 / 4$, then $V_{1}$ is ............ $m^3$
- 6In an isochoric process if ${T_1} = {27^o}C$ and ${T_2} = {127^o}C,$ then ${P_1}/{P_2}$ will be equal toView Solution
- 7A cyclic process $ABCD$ is shown in the $p-V$ diagram. Which of the following curves represents the same process if $BC \& DA$ are isothermal processesView Solution
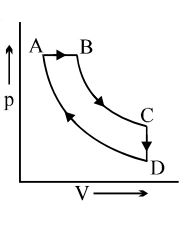
- 8The value of $\eta$ may lie betweenView Solution
- 9Match the thermodynamic processes taking place in a system with the correct conditions. In the table: $\Delta Q$ is the heat supplied, $\Delta W$ is the work done and $\Delta U$ is change in internal energy of the systemView Solution
Process Condition $(I)$ Adiabatic $(A)\; \Delta W =0$ $(II)$ Isothermal $(B)\; \Delta Q=0$ $(III)$ Isochoric $(C)\; \Delta U \neq 0, \Delta W \neq 0 \Delta Q \neq 0$ $(IV)$ Isobaric $(D)\; \Delta U =0$ - 10One mole of an ideal monoatomic gas undergoes the following four reversible processes:View Solution
Step $1$ It is first compressed adiabatically from volume $V_{1}$ to $1 \;m ^{3}$.
Step $2$ Then expanded isothermally to volume $10 \;m ^{3}$.
Step $3$ Then expanded adiabatically to volume $V _{3}$.
Step $4$ Then compressed isothermally to volume $V_{1}$. If the efficiency of the above cycle is $3 / 4$, then $V_{1}$ is ............ $m^3$
