In an isochoric process if ${T_1} = {27^o}C$ and ${T_2} = {127^o}C,$ then ${P_1}/{P_2}$ will be equal to
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
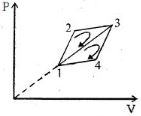
- 1The efficiency of a thermodynamic cycle $1-2-3- 1 ($see picture$)$ is $20\%$ and for another thermodynamic cycle $1 - 3-4 - 1$ efficiency is equal to $10\%$. Determine the efficiency $\eta ($ in $\%)$ of the thermodynamic cycle $1-2-3-4- 1.$The gas is assumed to be idealView Solution
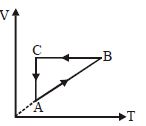
- 2A cyclic process $ABCA$ is shown in the $V-T$ diagram. Process on the $P-V$ diagram isView Solution
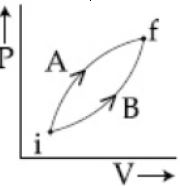
- 3View SolutionIn the adiabatic compression, the decrease in volume is associated with
- 4Following figure shows two processes $A$ and $B$ for a gas. If $\Delta Q_A$ and $\Delta Q_B$ are the amount of heat absorbed by the system in two cases, and $\Delta U_A$ and $\Delta U_B$ are changes in internal energies, respectively, thenView Solution
- 5A vessel containing $5\, litres$ of a gas at $0.8 \,pa$ pressure is connected to an evacuated vessel of volume $3$ litres. The resultant pressure inside will be ...... $pa$ (assuming whole system to be isolated)View Solution
- 6One mole of an ideal gas expands at a constant temperature of $300 \,K$ from an initial volume of $10\, litres$ to a final volume of $20\, litres$. The work done in expanding the gas is ...... $J.$ $(R = 8.31 J/mole-K)$View Solution
- 7A gas is suddenly compressed to one fourth of its original volume. What will be its final pressure, if its initial pressure is $P$View Solution
- 8A Car not engine whose low temperate reservoir is at $7\,^oC$ has an efficiency of $50\%$ . It is desired to increase the efficiency to $70\%$ . By how many degrees should the temperature of the high temperature reservoir be increased .... $K$View Solution
- 9Work done in the given $P-V$ diagram in the cyclic process isView Solution
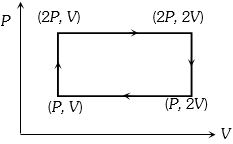
- 10A Carnot engine operates between two reservoirs of temperatures $900\; \mathrm{K}$ and $300 \;\mathrm{K}$ The engine performs $1200\; \mathrm{J}$ of work per cycle. The heat energy (in $\mathrm{J}$ ) delivered by the engine to the low temperature reservoir, in a cycle. isView Solution
