$300 \,cal$. of heat is given to a heat engine and it rejects $225 \,cal$. of heat. If source temperature is $227^{\circ} C$, then the temperature of sink will be____${ }^{\circ} C$.
JEE MAIN 2022, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A diatomic ideal gas is used in a Carnot engine as the working substance. If during the adiabatic expansion part of the cycle the volume of the gas increases from $V$ to $32\ V$, the efficiency of the engine isView Solution
- 2A carnot engine is made to work between $200\,^oC$ and $0\,^oC$ first and then between $0\,^oC$ and $-200\,^oC$. The ratio of efficiencies $\left( {\frac{{{\eta _2}}}{{{\eta _1}}}} \right)$ of the engine in two cases isView Solution
- 3View SolutionCan two isothermal curves cut each other
- 4One mole of an ideal gas is taken from a to $b$ along two paths denoted by the solid and the dashed lines as shown in the graph below. If the work done along the solid line path is $\mathrm{w}_{\mathrm{s}}$ and that along the dotted line path is $w_d$, then the integer closest to the ratio $w_d / w_5$ isView Solution
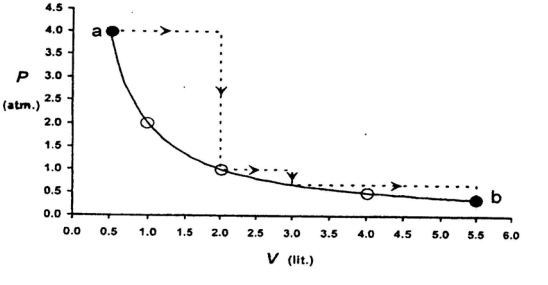
- 5A diatomic gas initially at $18^o C$ is compressed adiabatically to one-eighth of its original volume. The temperature after compression will beView Solution
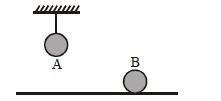
- 6Two identical balls, $A$ and $B$ , of uniform composition and initially at the same temperature, each absorb exactly the same amount of heat. $A$ is hanging down from the ceiling while $B$ rests on the horizontal floor in the same room. Assuming no subsequent heat loss by the balls, which of the following statements is correct about their final temperatures, $T_A$ and $T_B$ , once the balls have reached their final state?View Solution
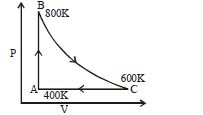
- 7One mole of diatomic ideal gas undergoes a cyclic process $ABC$ as shown in figure. The process $BC$ is adiabatic. The temperatures at $A, B$ and $C$ are $400\,K, 800\,K$ and $600\,K$ respectively. Choose the correct statementView Solution
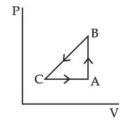
- 8A sample of an ideal gas is taken through the cyclic process $ABCA$ as shown in figure. It absorbs, $40\,J$ of heat during the part $A B$, no heat during $BC$ and rejects $60\,J$ of heat during $CA$. $A$ work $50\,J$ is done on the gas during the part $BC$. The internal energy of the gas at $A$ is $1560\,J$. The work done by the gas during the part $CA$ is.............$J$View Solution
- 9$n-$ $moles$ of an ideal gas with constant volume heat capacity $C_v$ undergo an isobaric expansion by certain volume. The ratio of the work done in the process, to the heat supplied isView Solution
- 10View SolutionFirst law of thermodynamics is a special case of
