The volume of an ideal gas $(\gamma=1.5)$ is changed adiabatically from $5$ litres to $4$ litres. The ratio of initial pressure to final pressure is:
JEE MAIN 2024, Diffcult
For Adiabatic process
$P_i V_i=P_f V_f^\gamma$
$P_i(5)^{1.5}=P_f(4)^{1.5}$
$\frac{P_i}{P_f}=\left(\frac{4}{5}\right)^{\frac{3}{2}}=\frac{4}{5} \cdot\left(\frac{4}{5}\right)^{\frac{1}{2}} \Rightarrow \frac{8}{5 \sqrt{5}}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Consider a carnot's cycle operating between $T_1 = 500\,K$ and $T_2 = 300\,K$ producing $1\,kJ$ of mechanical work per cycle. Find the heat transferred to the engine by the reservoirs .... $J$View Solution
- 2Choose the incorrect statement from the followingView Solution
$S1:$ The efficiency of a heat engine can be $1,$ but the coefficient of performance of a refrigerator can never be infinity.
$S2:$ The first law of thermodynamics is basically the principle of conservation of energy.
$S3:$ The second law of thermodynamics does not allow several phenomena consistent with the first law.
$S4:$ A process,whose only result is to transfer heat from a colder to a hotter object, is impossible.
- 3The efficiency of carnot engine is $50\%$ and temperature of sink is $500\,K$ . If temperature of source is kept constant and its efficiency raised to $60\%$ , then the required temperature of the sink will be .... $K$View Solution
- 4A Carnot engine has efficiency $25\%$ . It operates between reservoirs of constant temperature with temperature difference of $80\,K$ . What is the temperature of low temperature reservoir ...... $^oC$View Solution
- 5In the following $P-V$ diagram two adiabatics cut two isothermals at temperatures $T_1$ and $T_2$ (fig.). The value of $\frac{{{V_a}}}{{{V_d}}}$ will beView Solution
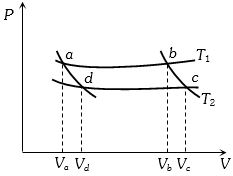
- 6View SolutionFirst law thermodynamics states that
- 7The temperature of a hypothetical gas increases to $\sqrt 2 $ times when compressed adiabatically to half the volume. Its equation can be written asView Solution
- 8A tyre filled with air $({27^o}C,$ and $2$ atm) bursts, then what is temperature of air ....... $^oC$ $(\gamma = 1.5)$View Solution
- 9A vessel containing $5\, litres$ of a gas at $0.8 \,pa$ pressure is connected to an evacuated vessel of volume $3$ litres. The resultant pressure inside will be ...... $pa$ (assuming whole system to be isolated)View Solution
- 10An ideal heat engine exhausting heat at $77\,^oC$. To have a $30\%$ efficiency. It must take heat at...... $^oC$View Solution
