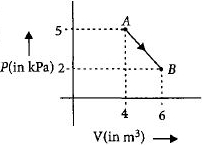
One mole of an ideal diatomic gas undergoes a transition from $A$ to $B$ along a path $AB$ as shown in the figure.
The change in internal energy of the gas during the transition is ............$\;kJ$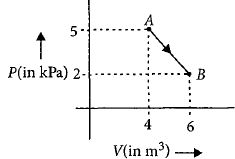
AIPMT 2015, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
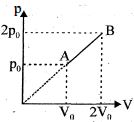
- 1An enclosed one mole of a monoatomic gas is taken through a process $A$ to $B$ as shown in figure. The molar heat capacity of the gas for this process isView Solution
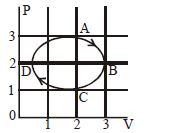
- 2The figure shows the $P-V$ plot of an ideal gas taken through a cycle $ABCDA.$ The part $ABC$ is a semicircle and $CDA$ is half of an ellipse. Then,View Solution
- 3A diatomic gas undergoes a process represented by $PV ^{1.3}=$ constant. Choose the incorrect statementView Solution
- 4$Assertion :$ In adiabatic compression, the internal energy and temperature of the system get decreased.View Solution
$Reason :$ The adiabatic compression is a slow process. - 5View SolutionIrreversible process is
- 6An ideal gas expands according to the law $P^2 V=$ constant. The internal energy of the gasView Solution
- 7A refrigerator consumes an average $35\, {W}$ power to operate between temperature $-10^{\circ} {C}$ to $25^{\circ} {C}$. If there is no loss of energy then how much average heat per second does it transfer? (in ${J} / {s}$)View Solution
- 8For adiabatic processes $\left( {\gamma = \frac{{{C_p}}}{{{C_v}}}} \right)$View Solution
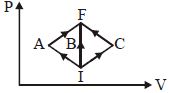
- 9In the $P-V$ diagram, $I$ is the initial state and $F$ is the final state. The gas goes from $I$ to $F$ by $(i)\,IAF,\,\,(ii)\,IBF,\,\,(iii) ICF$. The heat absorbed by the gas isView Solution
- 10Carnot cycle is plotted in $P-V$ graph. Which portion represents an isothermal expansion?View Solution