A Carnot engine take $5000 \,k\,cal$ of heat from a reservoir at $727\,^{\circ}C$ and gives heat to a $\operatorname{sink}$ at $127\,^{\circ}C$. The work done by the engine is $.......... \times 10^{6}\,J$
JEE MAIN 2022, Medium
$L=\frac{W D}{Q_{H}}$
$\Rightarrow WD = Q _{ H }\left(1-\frac{ T _{ L }}{ T _{ H }}\right)$
$=5 \times 10^{3}\left(1-\frac{400}{1000}\right)$
$=3000\, kcal$
$=12.6 \times 10^{6} J$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
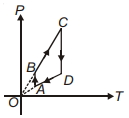
- 1A $P-T$ graph is shown for a cyclic process. Select correct statement regarding thisView Solution
- 2View SolutionIn adiabatic expansion
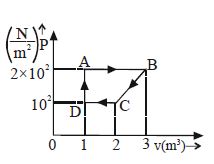
- 3A cyclic process is shown in figure. Work done during isobaric expansion is ...... $J$View Solution
- 4A gas mixture consists of $8$ moles of argon and $6$ moles of oxygen at temperature $T$. Neglecting all vibrational modes, the total internal energy of the system isView Solution
- 5A closed vessel contains $0.1$ mole of a monoatomic ideal gas at $200\, K$. If $0.05$ mole of the same gas at $400\, K$ is added to it, the final equilibrium temperature (in $K$ ) of the gas in the vessel will be closed toView Solution
- 6In a carnot engine, the temperature of reservoir is $527^{\circ} C$ and that of $\operatorname{sink}$ is $200 \; K$. If the workdone by the engine when it transfers heat from reservoir to sink is $12000 \; kJ$, the quantity of heat absorbed by the engine from reservoir is $\times 10^{6} \; J$View Solution
- 7A diatomic gas undergoes a process represented by $PV ^{1.3}=$ constant. Choose the incorrect statementView Solution
- 8View SolutionIn an isothermal process the volume of an ideal gas is halved. One can say that
- 9An ideal heat engine working between temperature $T_1$ and $T_2 $ has an efficiency $\eta$, the new efficiency if both the source and sink temperature are doubled, will beView Solution
- 10Determine efficiency of carnot cycle if in adiabatic expansion volume $3$ times of initial value and $\gamma =1.5$View Solution
