A Carnot engine working between $300\,K$ and $600\,K$ has work output of $800\, J$ per cycle. What is amount of heat energy supplied to the engine from source per cycle...... $J/cycle$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
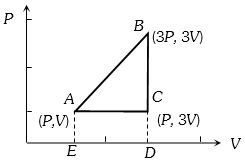
- 1One mole of an ideal monoatomic gas is taken along the path $ABCA$ as shown in the $PV$ diagram. The maximum temperature attained by the gas along the path $BC$ is given byView Solution
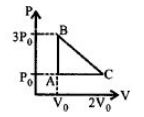
- 2View SolutionThe work done in an adiabatic change in a gas depends only on
- 3The gas law $\frac{{PV}}{T} = $ constant is true forView Solution
- 4An ideal gas follows a process described by $p V^2=C$ from $\left(p_1, V_1, T_1\right)$ to $\left(p_2, V_2, T_2\right)$ and $C$ is a constant. Then,View Solution
- 5A cyclic process $ABCD$ is shown in the $p-V$ diagram. Which of the following curves represents the same process if $BC \& DA$ are isothermal processesView Solution
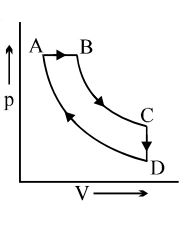
- 6View SolutionIn the following indicator diagram, the net amount of work done will be
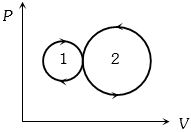
- 7During an adiabatic process, if the pressure of a gas is found to be proportional to the cube of its absolute temperature, then the ratio of $\frac{C_p}{C_V}$ for the gas is:View Solution
- 8An ideal gas follows a process $PT =$ constant. The correct graph between pressure $\&$ volume isView Solution
- 9A sample of an ideal gas is taken through the cyclic process $abca$ as shown in the figure. The change in the internal energy of the gas along the path $ca$ is $-180\, J$. The gas absorbs $250\, J$ of heat along the path $ab$ and $60\, J$ along the path $bc$. The work done by the gas along the path $abc$ is ..... $J$View Solution
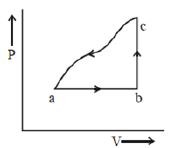
- 10An ideal gas is taken around $ABCA$ as shown in the above $P-V$ diagram. The work done during a cycle isView Solution