The gas law $\frac{{PV}}{T} = $ constant is true for
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
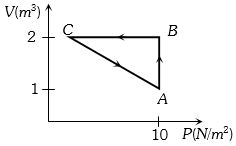
- 1An ideal gas is taken through the cycle $A → B → C → A$, as shown in the figure. If the net heat supplied to the gas in the cycle is $5 \ J$, the work done by the gas in the process $C → A$ is ....... $J$View Solution
- 2The value of $\eta$ may lie betweenView Solution
- 3One mole of an ideal gas expands at a constant temperature of $300 \,K$ from an initial volume of $10\, litres$ to a final volume of $20\, litres$. The work done in expanding the gas is ...... $J.$ $(R = 8.31 J/mole-K)$View Solution
- 4Which of the accompanying $PV$, diagrams best represents an isothermal processView Solution
- 5One mole of ${O_2}$ gas having a volume equal to $22.4$ litres at ${0^o}C$ and $1$ atmospheric pressure in compressed isothermally so that its volume reduces to $11.2$ litres. The work done in this process is ...... $J$View Solution
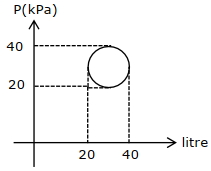
- 6In the reported figure, heat energy absorbed by a system in going through a cyclic process is $......\,\pi J$View Solution
- 7View SolutionThe ratio of work done by an ideal monoatomic gas to the heat supplied to it in an isobaric process is
- 8A gas is suddenly compressed to one fourth of its original volume. What will be its final pressure, if its initial pressure is $P$View Solution
- 9An ideal gas follows a process $PT =$ constant. The correct graph between pressure $\&$ volume isView Solution
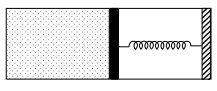
- 10An ideal monoatomic gas is confined in a horizontal cylinder by a spring loaded piston (as shown in the figure). Initially the gas is at temperature $T _1$, pressure $P_1$ and volume $V_1$ and the spring is in its relaxed state. The gas is then heated very slowly to temperature $T_2$, pressure $P _2$ and volume $V _2$. During this process the piston moves out by a distance $x$. Ignoring the friction between the piston and the cylinder, the correct statement$(s)$ is(are)View Solution
$(A)$ If $V_2=2 V_1$ and $T_2=3 T_1$, then the energy stored in the spring is $\frac{1}{4} P_1 V_1$
$(B)$ If $V_2=2 V_1$ and $T_2=3 T_1$, then the change in internal energy is $3 P_1 V_1$
$(C)$ If $V_2=3 V_1$ and $T_2=4 T_1$, then the work done by the gas is $\frac{7}{3} P_1 V_1$
$(D)$ If $V_2=3 V_1$ and $T_2=4 T_1$, then the heat supplied to the gas is $\frac{17}{6} P_1 V_1$