If $\gamma = 2.5$ and volume is equal to $\frac{1}{8}$ times to the initial volume then pressure $P' $ is equal to (Initial pressure $= P$)
Medium
(c)$\frac{{{P_2}}}{{{P_1}}} = {\left( {\frac{{{V_1}}}{{{V_2}}}} \right)^\gamma } \Rightarrow \frac{{P'}}{P} = {(8)^{5/2}} \Rightarrow P' = P \times {(2)^{15/2}}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
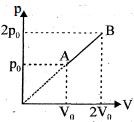
- 1An enclosed one mole of a monoatomic gas is taken through a process $A$ to $B$ as shown in figure. The molar heat capacity of the gas for this process isView Solution
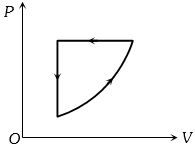
- 2For one complete cycle of a thermodynamic process on a gas as shown in the $P-V$ diagram, Which of following is correctView Solution
- 3A diatomic gas $(\gamma=1.4)$ does $400 J$ of work when it is expanded isobarically. The heat given to the gas in the process is ............ $J$View Solution
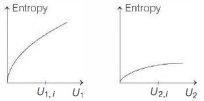
- 4Graphs below show the entropy versus energy $U$ of two systems $1$ and $2$ at constant volume. The initial energies of the systems are indicated by $U_{1, i}$ and $U_{2, i}$, respectively. Graphs are drawn to the same scale. The systems are then brought into thermal contact with each other. Assume that, at all times the combined energy of the two systems remains constant. Choose the most appropriate option indicating the energies of the two systems and the total entropy after they achieve the equilibrium.View Solution
- 5In a carnot engine, the temperature of reservoir is $527^{\circ} C$ and that of $\operatorname{sink}$ is $200 \; K$. If the workdone by the engine when it transfers heat from reservoir to sink is $12000 \; kJ$, the quantity of heat absorbed by the engine from reservoir is $\times 10^{6} \; J$View Solution
- 6Consider $1 \,kg$ of liquid water undergoing change in phase to water vapour at $100^{\circ} C$. At $100^{\circ} C$, the vapour pressure is $1.01 \times 10^5 \,N - m ^2$ and the latent heat of vaporization is $22.6 \times 10^5 \,Jkg ^{-1}$. The density of liquid water is $10^3 \,kg m ^{-3}$ and that of vapour is $\frac{1}{1.8} \,kg m ^{-3}$. The change in internal energy in this phase change is nearly ............ $\,J kg ^{-1}$View Solution
- 7The volume of air increases by $5\%$ in its adiabatic expansion. The percentage decrease in its pressure will be ...... $\%$View Solution
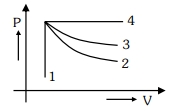
- 8An ideal gas undergoes four different processes from the same initial state as shown in the figure below. Those processes are adiabatic, isothermal, isobaric and isochoric. The curve which represents the adiabatic process among $1,2,3$ and $4$ isView Solution
- 9A tyre filled with air $({27^o}C,$ and $2$ atm) bursts, then what is temperature of air ....... $^oC$ $(\gamma = 1.5)$View Solution
- 10View SolutionIn isothermic process, which statement is wrong
