A closed vessel contains $8\,gm$ of oxygen and $7\,gm$ of nitrogen. The total pressure is $10\, atm$ at a given temperature. If now oxygen is absorbed by introducing a suitable absorbent the pressure of the remaining gas in atm will be
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Assertion $(A):$ The total translational kinetic energy of all the molecules of a given mass of an ideal gas is $1.5$ times the product of its pressure and volume.View Solution
Reason $(R):$ The molecules of gas collide with each other and the velocities of the molecules change due to the collision.
- 2View SolutionAccording to kinetic theory of gases,
- 3The temperature $(T)$ of one mole of an ideal gas varies with its volume $(V)$ as $T=-\alpha V^3+\beta V^2$, where $\alpha$ and $\beta$ are positive constants. The maximum pressure of gas during this process is ............View Solution
- 4A vessel contains $1$ mole of $O_2$ gas (molar mass $32$) at a temperature $T$. The pressure of the gas is $P$. An identical vessel containing one mole of $He$ gas (molar mass $4$) at a temperature $2T$ has a pressure ofView Solution
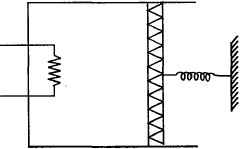
- 5An ideal monoatomic gas is confined in a cylinder by a spring loaded piston of cross section $8.0\times10^{-3}\, m^2$ . Initially the gas is at $300\, K$ and occupies a volume of $2.4\times10^{-3}\, m^3$ and the spring is in its relaxed state as shown in figure. The gas is heated by a small heater until the piston moves out slowly by $0.1\, m$. The force constant of the spring is $8000\, N/m$ and the atmospheric pressure is $1.0\times10^5\, N/m^2$ . The cylinder and the piston are thermally insulated. The piston and the spring are massless and there is no friction between the piston and the cylinder. The final temperature of the gas will be: (Neglect the heat loss through the lead wires of the heater . The heat capacity of the heater coil is also negligible)View Solution
- 6One mole of an ideal gas $\left( {\frac{{{C_P}}}{{{C_V}}}\, = \gamma } \right)$ heated by law $P=\alpha V$ where $P$ is pressure of gas, $V$ is volume, $\alpha$ is a constant what is the heat capacity of gas in the process-View Solution
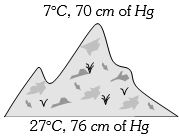
- 7At the top of a mountain a thermometer reads $7°C$ and a barometer reads $70\, cm$ of $Hg.$ At the bottom of the mountain these read $27°C$ and $76 \,cm$ of $Hg$ respectively. Comparison of density of air at the top with that of bottom isView Solution
- 8The kinetic energy of one gm-mole of a gas at normal temperature and pressure is $(R = 8.31 J/Mole-K)$View Solution
- 9At a given temperature the ratio of $r.m.s.$ velocities of hydrogen molecule and helium atom will beView Solution
- 10Given below are two statements :View Solution
Statement $(I)$ : The mean free path of gas molecules is inversely proportional to square of molecular diameter.
Statement $(II)$ : Average kinetic energy of gas molecules is directly proportional to absolute temperature of gas.
In the light of the above statements, choose the correct answer from the option given below:
