Let $\gamma_1$ be the ratio of molar specific heat at constant pressure and molar specific heat at constant volume of a monoatomic gas and $\gamma_2$ be the similar ratio of diatomic gas. Considering the diatomic gas molecule as a rigid rotator, the ratio, $\frac{\gamma_1}{\gamma_2}$ is
JEE MAIN 2023, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionEvery gas (real gas) behaves as an ideal gas
- 2A vessel of volume $0.02 \,\,m^3$ contains a mixture of hydrogen and helium at $20\,^oC$ and $2$ atmospheric pressure. The mass of mixture is $5\,\, gms$. Find the ratio of mass of hydrogen to that of helium in the mixture.View Solution
- 3Volume-temperature graph at atmospheric pressure for a monoatomic gas $(V\,{\rm{in }}{m^3},\,\,T\,{\rm{in}}{{\rm{ }}^{\rm{o}}}C)$ isView Solution
- 4$Assertion :$ Mean free path of a gas molecules varies inversely as density of the gas.View Solution
$Reason :$ Mean free path varies inversely as pressure of the gas. - 5The average translational kinetic energy of a hydrogen gas molecules at $NTP$ will beView Solution
[Boltzmann’s constant ${k_B} = 1.38 \times {10^{ - 23}}J/K]$
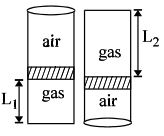
- 6An ideal gas is trapped inside a test tube of cross-sectional area $20 \times 10^{-6} \,\,m^2$ as shown in the figure. The gas occupies a height $L_1$ at the bottom of the tube and is separated from air at atmospheric pressure by a mercury column of mass $0.002\,\, kg$. If the tube is quickly turned isothermally, upside down so that $L_2$ mercury column encloses the gas from below. The gas now occupies height $L_1$ in the tube. The ratio $L_1$ is [Take atmospheric pressure $= 10^5 Nm^{-2}]$View Solution
- 7At $NTP$ the mass of one litre of air is $1.293\, gm.$ The value of specific gas constant will be ...... $J/K-gm$View Solution
- 8View SolutionWhen volume of system is increased two times and temperature is decreased half of its initial temperature, then pressure becomes ...... times
- 9In Vander Waal's equation $\left[ {P + \frac{a}{{{V^2}}}} \right]\,(V - b) = RT,$ the dimensions of $a$ areView Solution
- 10A perfect gas at $27°C$ is heated at constant pressure so as to triple its volume. The temperature of the gas will be ...... $^oC$View Solution
