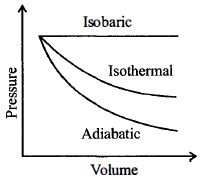
A gas is compressed adiabatically, which one of the following statement is NOT true.
JEE MAIN 2023, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
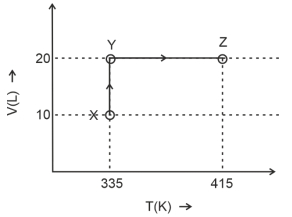
- 1Consider the following volume-temperature $( V - T )$ diagram for the expansion of $5$ moles of an ideal monoatomic gas.View Solution
Considering only $P-V$ work is involved, the total change in enthalpy (in Joule) for the transformation of state in the sequence $X \rightarrow Y \rightarrow Z$ is $\qquad$
[Use the given data: Molar heat capacity of the gas for the given temperature range, $C _{ v , m }=12 J K ^{-1} mol ^{-1}$ and gas constant, $R =8.3 J K ^{-1} mol ^{-1}$ ]
- 2View Solution“Heat cannot by itself flow from a body at lower temperature to a body at higher temperature” is a statement or consequence of
- 3Efficiency of Carnot engine is $100\%$ ifView Solution
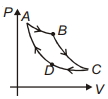
- 4Carnot cycle is plotted in $P-V$ graph. Which portion represents an isothermal expansion?View Solution
- 5The temperature inside a refrigerator is $t_2 \,^o C$ and the room temperature is $t_1\,^o C.$ The amount of heat delivered to the room for each joule of electrical energy consumed ideally will beView Solution
- 6View SolutionIrreversible process is
- 7One mole of an ideal gas at $300 \mathrm{~K}$ in thermal contact with surroundings expands isothermally from $1.0 \mathrm{~L}$ to $2.0 \mathrm{~L}$ against a constant pressure of $3.0 \mathrm{~atm}$. In this process, the change in entropy of surroundings $\left(\Delta S_{\text {surr }}\right)$ in $\mathrm{J} \mathrm{K}^{-1}$ is $(1 \mathrm{~L} \mathrm{~atm}=101.3 \mathrm{~J})$View Solution
- 8View SolutionEntropy of a thermodynamic system does not change when this system is used for
- 9An ideal heat engine exhausting heat at $77\,^oC$. To have a $30\%$ efficiency. It must take heat at...... $^oC$View Solution
- 10A sample of gas expands from $V_1$ to $V _2$. In which of the following, the work done will be greatest ?View Solution