A thermodynamic system is taken from state $A$ to $B$ along $ACB$ and is brought back to $A$ along $BDA$ as shown in the $PV$ diagram. The net work done during the complete cycle is given by the area
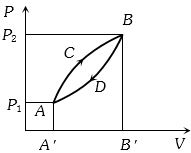

AIPMT 1992, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A heat engine operates between a cold reservoir at temperature ${T}_{2}=400\, {K}$ and a hot reservoir at temperature ${T}_{1} .$ It takes $300 \,{J}$ of heat from the hot reservoir and delivers $240\, {J}$ of heat to the cold reservoir in a cycle. The minimum temperature of the hot reservoir has to be $....{K}$View Solution
- 2The adjoining figure shows the $P-V$ diagram for a fixed mass of an ideal gas undergoing cyclic process. $AB$ represents isothermal process and $CA$ represents isochoric process.Which of the graph shown in the following figures represents the $P-T$ diagram of the cyclic process ?View Solution
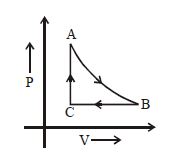
- 3A Carnot engine, having an efficiency of $\eta = 1/10$ as heat engine, is used as a refrigerator. If the work done on the system is $10\ J$, the amount of energy absorbed from the reservoir at lower temperature is ....... $J$View Solution
- 4View SolutionThe state of an ideal gas was changed isobarically. The graph depicts three such isobaric lines. Which of the following is true about the pressures of the gas?
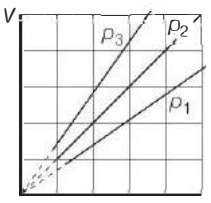
- 5View SolutionOne mole of a monoatomic gas and one mole of a diatomic gas are initially in the same state. Both gases are expanded isothermally and then adiabatically, such that they acquire the same final state. Choose the correct statement.
- 6Match List$-I$ with List$-II$View Solution
List$-I$ List$-II$ $(a)$ Isothermal $(i)$ Pressure constant $(b)$ Isochoric $(ii)$ Temperature constant $(c)$ Adiabatic $(iii)$ Volume constant $(d)$ Isobaric $(iv)$ Heat content is constant Choose the correct answer from the options given below
- 7$PV$ curve for the process whose $VT$ curve isView Solution
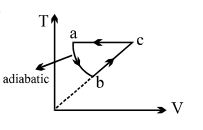
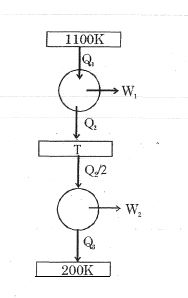
- 8Consider the given series combination of carnot cycles. If $W_1 = W_2$ then the value of $T$ is ...... $K$ (all temperatures are maintained at their respective values)View Solution
- 9During an adiabatic process, the pressure of a gas is found to be proportional to the cube of its temperature. The ratio of $\frac{{{C_P}}}{{{C_V}}}$ for the gas isView Solution
- 10View SolutionThe work done in which of the following processes is zero
