For an ideal gas, the internal energy is given by $U=5 p V / 2+C$, where $C$ is a constant. The equation of the adiabats in the $p V$-plane will be
KVPY 2018, Advanced
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A gas mixture consists of $8$ moles of argon and $6$ moles of oxygen at temperature $T$. Neglecting all vibrational modes, the total internal energy of the system isView Solution
- 2A reversible engine has an efficiency of $\frac{1}{4}$. If the temperature of the sink is reduced by $58^{\circ} {C}$, its efficiency becomes double. Calculate the temperature of the sink. (In $^{\circ} {C}$)View Solution
- 3The pressure of an ideal gas varies with volume as $P = \alpha V,$ where $\alpha $ is a constant. One mole of the gas is allowed to undergo expansion such that its volume becomes $'m'$ times its initial volume. The work done by the gas in the process isView Solution
- 4A gas is enclosed in a cylinder with a movable frictionless piston. Its initikl thermodynamic state at pressure $P_i=10^5 \mathrm{~Pa}$ and volume $V_i=10^{-3} \mathrm{~m}^3$ chanıes to i final state at $P_f=(1 / 32) \times 10^5 \mathrm{~Pa}$ and $V_f=8 \times 10^{-3} \mathrm{~m}^3$ in an adiabatic quasi-static process, such that $P^3 V^5=$ constant. Consider another thermodynamic process that brings the system from the same initial state to the same final state in two steps: an isobaric expansion at $P$, followed by an isochoric (isovolumetric) process at volume $V_f$. The amount of heat supplied to the system in the two-step process is approximatelyView Solution
- 5$5.6$ $liter$ of helium gas at $STP$ is adiabatically compressed to $0.7$ $liter$. Taking the initial temperature to be $T_1$, the work done in the process isView Solution
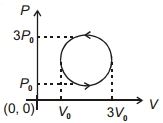
- 6View SolutionWork done in the cyclic process shown in figure is ...........
- 7Air is filled in a motor tube at ${27^o}C$ and at a pressure of $8$ atmospheres. The tube suddenly bursts, then temperature of air is $[{\rm{Given}}\,\,\gamma \,{\rm{of}}\,{\rm{air}} = \,1.5]$View Solution
- 8View SolutionWhich of the following can be coefficient of performance of refrigerator?
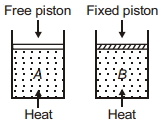
- 9Two cylinders contain same amount of ideal monatomic gas. Same amount of heat is given to two cylinders. If temperature rise in cylinder $A$ is $T_0$ then temperature rise in cylinder $B$ will be .........View Solution
- 10$Assertion :$ In free expansion of an ideal gas, the entropy increases.View Solution
$Reason :$ Entropy increases in all natural processes.
