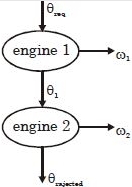
Suppose that two heat engines are connected in series, such that the heat released by the first engine is used as the heat absorbed by the second engine, as shown in figure. The efficiencies of the engines are $\in_1$ and $\in_2$, respectively. The net efficiency of the combination is given by :

Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The efficiency of an ideal heat engine working between the freezing point and boiling point of water, is ........ $\%$View Solution
- 2$List I$ describes thermodynamic processes in four different systems. $List II$ gives the magnitudes (either exactly or as a close approximation) of possible changes in the internal energy of the system due to the process.View Solution
$List-I$ $List-II$ ($I$) $10^{-3} kg$ of water at $100^{\circ} C$ is converted to steam at the same temperature, at a pressure of $10^5 Pa$. The volume of the system changes from $10^{-6} m ^3$ to $10^{-3} m ^3$ in the process. Latent heat of water $=2250 kJ / kg$. ($P$) $2 kJ$ ($II$) $0.2$ moles of a rigid diatomic ideal gas with volume $V$ at temperature $500 K$ undergoes an isobaric expansion to volume $3 V$. Assume $R=8.0 Jmol ^1 K^{-1}$. ($Q$) $7 kJ$ ($III$) On mole of a monatomic ideal gas is compressed adiabatically from volume $V=\frac{1}{3} m^3$ and pressure $2 kPa$ to volume $\frac{v}{8}$ ($R$) $4 kJ$ ($IV$) Three moles of a diatomic ideal gas whose molecules can vibrate, is given $9 kJ$ of heat and undergoes isobaric expansion. ($S$) $5 kJ$ ($T$) $3 kJ$ Which one of the following options is correct?
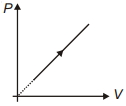
- 3View SolutionDuring the thermodynamic process shown in figure for an ideal gas
- 4An engineer claims to have made an engine delivering $10 kW$ power with fuel consumption of $1\,g\,{s^{ - 1}}$. The calorific value of fuel is $2k cal/g$. His claimView Solution
- 5View SolutionWhen heat in given to a gas in an isobaric process, then
- 6For a thermodynamic process $\delta Q = -50$ $calorie$ and $W = -20$ $calorie$ . If the initial internal energy is $-30$ $calorie$ then final internal energy will be ....... $calorie$View Solution
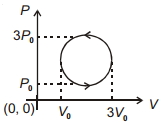
- 7View SolutionWork done in the cyclic process shown in figure is ...........
- 8View SolutionWhen heat in given to a gas in an isobaric process, then
- 9View SolutionAn ideal diatomic gas is heated at constant pressure. The ratio of the work done to the heat supplied is
- 10An ideal gas has volume ${V_0}$ at ${27^o}C.$ It is heated at constant pressure so that its volume becomes $2{V_0}.$ The final temperature isView Solution