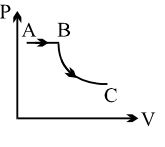
An ideal gas undergoes a cyclic process as shown in diagram. The net work done by the gas in the cycle is
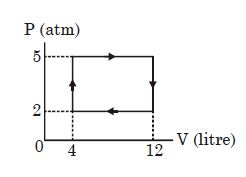

Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A gas is taken through the cycle $A\to B\to C\to A$ as shown. What is the net work done by the gas ...... $J$ $?$View Solution
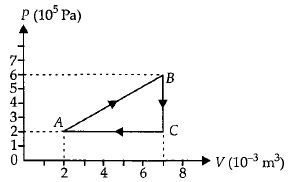
- 2View SolutionThe slopes of isothermal and adiabatic curves are related as
- 3View SolutionWhich relation is correct for isometric process
- 4An ideal gas is taken around $ABCA$ as shown in the above $P-V$ diagram. The work done during a cycle isView Solution
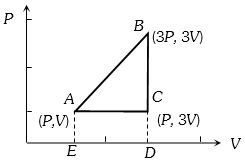
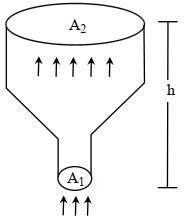
- 5An ideal gas of density $\rho=0.2 kg m ^{-3}$ enters a chimney of height $h$ at the rate of $\alpha=0.8 kg s ^{-1}$ from its lower end, and escapes through the upper end as shown in the figure. The cross-sectional area of the lower end is $A_1=0.1 m ^2$ and the upper end is $A_2=0.4 m ^2$. The pressure and the temperature of the gas at the lower end are $600 Pa$ and $300 K$, respectively, while its temperature at the upper end is $150 K$. The chimney is heat insulated so that the gas undergoes adiabatic expansion. Take $g=10 ms ^{-2}$ and the ratio of specific heats of the gas $\gamma=2$. Ignore atmospheric pressure.View Solution
Which of the following statement($s$) is(are) correct?
- 6When an ideal gas in a cylinder was compressed isothermally by a piston, the work done on the gas was found to be $1.5 \times {10^4}\;joules$. During this process aboutView Solution
- 7The graph of pressure $(P)$ and volume $(V)$ according to $PV^n = C$, here $n = 1.4$View Solution
- 8View SolutionA process is shown in the diagram. Which of the following curves may represent the same process ?
- 9View SolutionWhen an ideal triatomic non-linear gas is heated at constant pressure, the fraction of the heat energy supplied which increases the internal energy of the gas is
- 10$P_i, V_i$ and $P_f$, $V_f$ are initial and final pressures and volumes of a gas in a thermodynamic process respectively. If $PV^n =$ constant, then the amount of work done isView Solution
