An ideal gas follows a process described by the equation $PV ^2= C$ from the initial $\left( P _1, V _1, T _1\right)$ to final $\left(P_2, V_2, T_2\right)$ thermodynamics states, where $C$ is a constant. Then
NEET 2022, Medium
$PV ^2= C$
$\Rightarrow \frac{ nRT }{ V } V ^2= C$
$\Rightarrow TV =\text { constant }$
$\therefore V _2 > V _1 \Rightarrow T _1 > T _2$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1If $\Delta U$ and $\Delta W$ represent the increase in internal energy and work done by the system respectively in a thermodynamical process, which of the following is true?View Solution
- 2A gas may expand either adiabatically or isothermally. A number of $P-V$ curves are drawn for the two processes over different range of pressure and volume. It will be found thatView Solution
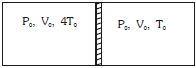
- 3Figure shows a cylindrical adiabatic container of total volume $2V_0$ divided into two equal parts by a conducting piston (which is free to move). Each part containing identical gas at pressure $P_0$ . Initially temperature of left and right part is $4T_0$ and $T_0$ respectively. An external force is applied on the piston to keep the piston at rest. Find the value of external force required when thermal equilibrium is reached. ( $A =$ Area of piston)View Solution
- 4An engine has an efficiency of $1/6.$ When the temperature of sink is reduced by $62^o C,$ its efficiency is doubled. Temperatures of the source is ....... $^oC$View Solution
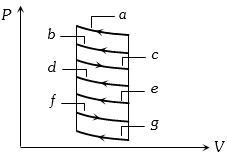
- 5The $P-V$ diagram shows seven curved paths (connected by vertical paths) that can be followed by a gas. Which two of them should be parts of a closed cycle if the net work done by the gas is to be at its maximum valueView Solution
- 6A piece of hot copper at $100^{\circ} C$ is plunged into a pond at $30^{\circ} C$. The copper cools down to $30^{\circ} C$, while the pond being huge stays at its initial temperature. Then,View Solution
- 7A thermally insulated rigid container contains an ideal gas heated by a filament of resistance $100 \,\Omega$ through a current of $1\,A$ for $5$ min then change in internal energy is...... $kJ$View Solution
- 8Efficiency of Carnot engine is $100\%$ ifView Solution
- 9An electric heater supplies heat to a system at a rate of $100\;W.$ If system performs work at a rate of $75$ Joules per second. At what rate (in $J/s$) is the internal energy increasing?View Solution
- 10An ideal gas is subjected to an isothermal expansion such that its volume changes from $V_i$ to $V_f$ and pressure from $P_i$ to $P_f$. The work done on the gas is :View Solution
