If $300 ml$ of a gas at ${27^o}C$ is cooled to ${7^o}C$ at constant pressure, then its final volume will be ...... $ml$
Easy
(c) $V \propto T$ at constant pressure
==> $\frac{{{V_1}}}{{{V_2}}} = \frac{{{T_1}}}{{{T_2}}}$ ==> ${V_2} = \frac{{{V_1}{T_2}}}{{{T_1}}} = \frac{{300 \times 280}}{{300}} = 280\,ml.$
==> $\frac{{{V_1}}}{{{V_2}}} = \frac{{{T_1}}}{{{T_2}}}$ ==> ${V_2} = \frac{{{V_1}{T_2}}}{{{T_1}}} = \frac{{300 \times 280}}{{300}} = 280\,ml.$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The heat energy required to raise the temperature of $5\,moles$ of an ideal gas to $5\,K$ at constant pressure is $600\,J$ . How much heat (in $J$ ) is required to raise the same mass of the same gas to $5\,K$ at constant volume ? (Take $R = 8.3\,J/mole-^oK$ )View Solution
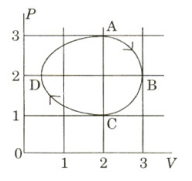
- 2The figure shows the $P-V$ plot of an ideal gas taken through a cycle $ABCDA$. The part $ABC$ is a semi-circle and $CDA$ is half of an ellipse. Then,View Solution
$(A)$ the process during the path $\mathrm{A} \rightarrow \mathrm{B}$ is isothermal
$(B)$ heat flows out of the gas during the path $\mathrm{B} \rightarrow \mathrm{C} \rightarrow \mathrm{D}$
$(C)$ work done during the path $\mathrm{A} \rightarrow \mathrm{B} \rightarrow \mathrm{C}$ is zero
$(D)$ positive work is done by the gas in the cycle $ABCDA$
- 3Match the thermodynamic processes taking place in a system with the correct conditions. In the table: $\Delta Q$ is the heat supplied, $\Delta W$ is the work done and $\Delta U$ is change in internal energy of the systemView Solution
Process Condition $(I)$ Adiabatic $(A)\; \Delta W =0$ $(II)$ Isothermal $(B)\; \Delta Q=0$ $(III)$ Isochoric $(C)\; \Delta U \neq 0, \Delta W \neq 0 \Delta Q \neq 0$ $(IV)$ Isobaric $(D)\; \Delta U =0$ - 4An electric appliance supplies $6000\, {J} / {min}$ heat to the system. If the system delivers a power of $90\, {W}$. How long (in $sec$) it would take to increase the internal energy by $2.5 \times 10^{3}\, {J}$ ?View Solution
- 5An ideal gas undergoes the process $1 \rightarrow 2$ as shown in the figure, the heat supplied and work done in the process is $\Delta \,\,Q$ and $\Delta \,\,W$ respectively. The ratio $\Delta \,\,Q :$ $\Delta \,\,W$ isView Solution
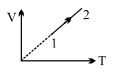
- 6View SolutionThe maximum possible efficiency of a heat engine is ...........
- 7View SolutionWhen an ideal diatomic gas is heated at constant pressure, the fraction of the heat energy supplied which increases the internal energy of the gas, is
- 8View SolutionIn the following figures heat is absorbed by the gas
- 9If a Carnot’s engine functions at source temperature $127^o C$ and at sink temperature $87^o C$, what is its efficiency ....... $\%$View Solution
- 10One mole of an ideal monoatomic gas is compressed isothermally in a rigid vessel to double its pressure at room temperature, $27\,^oC$.The work done on the gas will beView Solution
