$\mathrm{PV}^{\mathrm{y}}=\mathrm{constant} \ldots \ldots \ldots(1)$
As volume $=[(\text { mass }) /(\text { density })]$ i.e. $\quad \mathrm{V}=(\mathrm{m} / \mathrm{d})$
$\left(\mathrm{V}_{1} / \mathrm{V}_{2}\right)=\left(\mathrm{d}_{2} / \mathrm{d}_{1}\right) \ldots \ldots \ldots(2)$
From $(1),\left(P_{1} / P_{2}\right)=\left(V_{2} / V_{1}\right)^{Y}$
From $(2),\left(P_{1} / P_{2}\right)=\left(d_{1} / d_{2}\right)^{Y}$
Here $P_{1}=P, P_{2}=P^{\prime}, d_{1}=d, d_{2}=d^{\prime}$
Hence $\left(P / P^{1}\right)=\left(d / d^{\prime}\right)^{y}$
$\therefore\left(\mathrm{P} / \mathrm{P}^{1}\right)=(1 / 32)^{7 / 5}$
$\therefore\left(\mathrm{P} / \mathrm{P}^{1}\right)=\left[1 / 2^{\{5 \times(7 / 5)\}}\right]=\left(1 / 2^{7}\right)$
$\therefore P^{\prime}=2^{7} \cdot P$
$\therefore\left(P^{\prime} / P\right)=128$
Download our appand get started for free
Similar Questions
- 1View SolutionAn adiabatic process occurs at constant
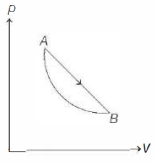
- 2An ideal gas is made to undergo a cycle depicted by the $p-V$ diagram given below. The curved line from $A$ to $B$ is an adiabat.Then,View Solution
- 3A diatomic gas, having $C_{p}=\frac{7}{2} R$ and $C _{ v }=\frac{5}{2} R ,$ is heated at constant pressure. The ratio $dU : dQ : dW :$View Solution
- 4In a mechanical refrigerator, the low temperature coils are at a temperature of $-23°C$ and the compressed gas in the condenser has a temperature of $27°C.$ The theoretical coefficient of performance isView Solution
- 5$List I$ describes thermodynamic processes in four different systems. $List II$ gives the magnitudes (either exactly or as a close approximation) of possible changes in the internal energy of the system due to the process.View Solution
$List-I$ $List-II$ ($I$) $10^{-3} kg$ of water at $100^{\circ} C$ is converted to steam at the same temperature, at a pressure of $10^5 Pa$. The volume of the system changes from $10^{-6} m ^3$ to $10^{-3} m ^3$ in the process. Latent heat of water $=2250 kJ / kg$. ($P$) $2 kJ$ ($II$) $0.2$ moles of a rigid diatomic ideal gas with volume $V$ at temperature $500 K$ undergoes an isobaric expansion to volume $3 V$. Assume $R=8.0 Jmol ^1 K^{-1}$. ($Q$) $7 kJ$ ($III$) On mole of a monatomic ideal gas is compressed adiabatically from volume $V=\frac{1}{3} m^3$ and pressure $2 kPa$ to volume $\frac{v}{8}$ ($R$) $4 kJ$ ($IV$) Three moles of a diatomic ideal gas whose molecules can vibrate, is given $9 kJ$ of heat and undergoes isobaric expansion. ($S$) $5 kJ$ ($T$) $3 kJ$ Which one of the following options is correct?
- 6In an adiabatic process, the state of a gas is changed from ${P_1},{V_1},{T_1} $ to ${P_2},{V_2},{T_2}$. Which of the following relation is correctView Solution
- 7A Carnot engine has efficiency of $50 \%$. If the temperature of sink is reduced by $40^{\circ} C$, its efficiency increases by $30 \%$. The temperature of the source will be$....K$View Solution
- 8Air is pumped into a balloon, of initial volume $V$ , until its diameter has doubled. If the atmospheric pressure is $p$ , what is the work done against the atmosphere ?View Solution
- 9A sample of gas expands from $V_1$ to $V _2$. In which of the following, the work done will be greatest ?View Solution
- 10The internal energy change in a system that has absorbed $2 \;k cal$ of heat and done $500 \;J $ of work is ...... $J$View Solution
