Acid catalysed hydration of alkenes except ethene leads to the formation of.
- Secondary or tertiary alcohol.
Alkenes react with water in the presence of acid as a catalyst to form alcohol. In case of unsymmetrical alkenes, OH is added to the carbon having fewer hydrogen atoms according to Markovnikov's rule.
Hence, acid catalyzed hydration of alkenes except ethene leads to the formation of secondary or tertiary alcohol. To obtain primary alcohol, hydroboration oxidation is used.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
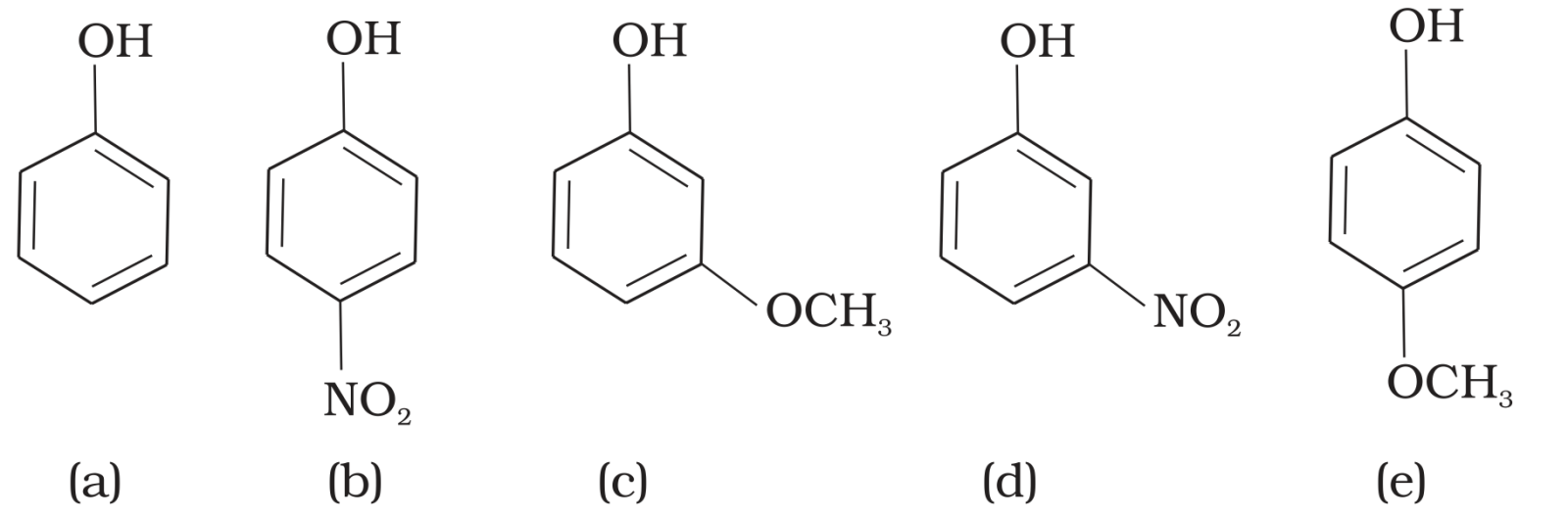
- 1The nature of $2, 4, 6-$trinitrophenol is$:$View Solution
- 2View SolutionGive the IUPAC name of the above compound.
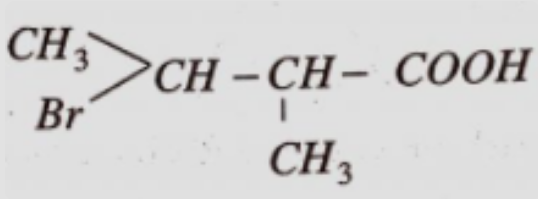
- 3View SolutionWhat is the functional group in alcohol?
- 4Give IUPAC name of the compound given below.View Solution
$\text{CH}_3-\text{CH}-\text{CH}_2-\text{CH}_2-\text{CH}-\text{CH}_3 \\ \ \ \ \ \ \ \ \ \ \ \ \ \ \ | \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ | \\ \ \ \ \ \ \ \ \ \ \ \ \ \ \text{Cl} \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \text{CH}$ - 5View SolutionWhich of the following is the functional group of an ether?
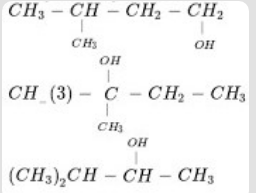
- 6View SolutionArrange the following alkanols 1, 2 and 3 in order of their reactivity towards acid catalyzed dehydration.
- 7View SolutionWhich of the following statement is not correct?
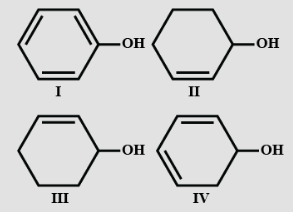
- 8View SolutionDehydration of the following in increasing order is:
- 9View SolutionIUPAC name of phenol is -
- 10View SolutionMark the correct order of decreasing acid strength of the following compounds.