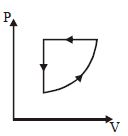
A sample of gas with $\gamma=1.5$ is taken through an adiabatic process in which the volume is compressed from $1200\, {cm}^{3}$ to $300\, {cm}^{3}$. If the initial pressure is $200\, {kPa}$. The absolute value of the workdone by the gas in the process $= \,..... J.$
JEE MAIN 2021, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1One mole of an ideal gas $(\gamma = 1.4)$ is adiabatically compressed so that its temperature rises from $27\,^oC$ to $35\,^oC$ . The change in the internal energy of the gas is .... $J$. (given $R = 8.3\,J/mole-K$ )View Solution
- 2In an isochoric process if ${T_1} = {27^o}C$ and ${T_2} = {127^o}C,$ then ${P_1}/{P_2}$ will be equal toView Solution
- 3$Assertion :$ In adiabatic compression, the internal energy and temperature of the system get decreased.View Solution
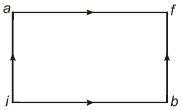
$Reason :$ The adiabatic compression is a slow process. - 4When a system is taken from state $i$ to state $f$ along the path $iaf$, it is found that $Q=50$ $cal$ and $W=20$ $cal$ Along the path $ibf\ Q = 36\ cal. \ W$ along the path $ibf$ is ....... $ cal$View Solution
- 5View SolutionIn a thermodynamic system working substance is ideal gas, its internal energy is in the form of
- 6An electric appliance supplies $6000\, {J} / {min}$ heat to the system. If the system delivers a power of $90\, {W}$. How long (in $sec$) it would take to increase the internal energy by $2.5 \times 10^{3}\, {J}$ ?View Solution
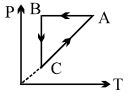
- 7A cyclic process $ABCA$ is shown in $PT$ diagram. When presented on $PV$, it wouldView Solution
- 8A certain amount of gas of volume $V$ at $27^{o}\,C$ temperature and pressure $2 \times 10^{7} \;Nm ^{-2}$ expands isothermally until its volume gets doubled. Later it expands adiabatically until its volume gets redoubled. The final pressure of the gas will be (Use $\gamma=1.5$ )View Solution
- 9View SolutionThe ratio of work done by an ideal monoatomic gas to the heat supplied to it in an isobaric process is
- 10For one complete cycle of a thermodynamic processes on a gas as shown in $P-V$ diagram. Which is trueView Solution