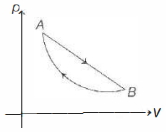
An ideal gas is made to undergo a cycle depicted by the $p-V$ diagram given below. The curved line from $A$ to $B$ is an adiabat.Then,

KVPY 2010, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1If $R =$ universal gas constant, the amount of heat needed to raise the temperature of $2$ mole of an ideal monoatomic gas from $273K$ to $373K$ when no work is done ...... $R$View Solution
- 2An ideal gas undergoes a polytropic given by equation $P V^n=$ constant. If molar heat capacity of gas during this process is arithmetic mean of its molar heat capacity at constant pressure and constant volume then value of $n$ is ..............View Solution
- 3An ideal gas is subjected to a thermodynamic process $PV^{2.5} = 0.40$ where $P$ is in $Pa$ and $V$ is in $m^3$.What is the slope of the $P-V$ curve with volume plotted against $x-$ axis at $V=1\, m^3$ ?View Solution
- 4A bicycle tyre is filled with air having pressure of $270\,kPa$ at $27^{\circ}\,C$. The approximate pressure of the air in the tyre when the temperature increases to $36^{\circ}\,C$ is $............kPa$View Solution
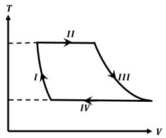
- 5One mole of a monatomic ideal gas undergoes a cyclic process as shown in the figure (where $V$ is the volume and $T$ is the temperature). Which of the statements below is (are) true?View Solution
(image)
$(A)$ Process $I$ is an isochoric process $(B)$ In process $II$, gas absorbs heat
$(C)$ In process $IV$, gas releases heat $(D)$ Processes $I$ and $III$ are $not$ isobaric
- 6One mole of an ideal gas $(\gamma = 1.4)$ is adiabatically compressed so that its temperature rises from $27\,^oC$ to $35\,^oC$ . The change in the internal energy of the gas is .... $J$. (given $R = 8.3\,J/mole-K$ )View Solution
- 7$List I$ describes thermodynamic processes in four different systems. $List II$ gives the magnitudes (either exactly or as a close approximation) of possible changes in the internal energy of the system due to the process.View Solution
$List-I$ $List-II$ ($I$) $10^{-3} kg$ of water at $100^{\circ} C$ is converted to steam at the same temperature, at a pressure of $10^5 Pa$. The volume of the system changes from $10^{-6} m ^3$ to $10^{-3} m ^3$ in the process. Latent heat of water $=2250 kJ / kg$. ($P$) $2 kJ$ ($II$) $0.2$ moles of a rigid diatomic ideal gas with volume $V$ at temperature $500 K$ undergoes an isobaric expansion to volume $3 V$. Assume $R=8.0 Jmol ^1 K^{-1}$. ($Q$) $7 kJ$ ($III$) On mole of a monatomic ideal gas is compressed adiabatically from volume $V=\frac{1}{3} m^3$ and pressure $2 kPa$ to volume $\frac{v}{8}$ ($R$) $4 kJ$ ($IV$) Three moles of a diatomic ideal gas whose molecules can vibrate, is given $9 kJ$ of heat and undergoes isobaric expansion. ($S$) $5 kJ$ ($T$) $3 kJ$ Which one of the following options is correct?
- 8A gas ($\gamma = 1.3)$ is enclosed in an insulated vessel fitted with insulating piston at a pressure of ${10^5}\,N/{m^2}$. On suddenly pressing the piston the volume is reduced to half the initial volume. The final pressure of the gas isView Solution
- 9A real gas within a closed chamber at $27^{\circ} \mathrm{C}$ undergoes the cyclic process as shown in figure. The gas obeys $P V^3=\mathrm{RT}$ equation for the path $A$ to $B$. The net work done in the complete cycle is (assuming $R=8 \mathrm{~J} / \mathrm{molK}$ ):View Solution
- 10An ideal gas is subjected to an isothermal expansion such that its volume changes from $V_i$ to $V_f$ and pressure from $P_i$ to $P_f$. The work done on the gas is :View Solution