An ideal gas heat engine operates in Carnot cycle between $227°C$ and $127°C.$ It absorbs $6 \times {10^4}$ cals of heat at higher temperature. Amount of heat converted to work is .........$ \times {10^4}\; cal$
AIPMT 2005, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A thermodynamic system is taken through cyclic process. The total work done in the process is $.........\,J$View Solution
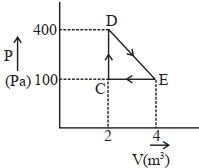
- 2Suppose ideal gas equation follows $V{P^3}$= constant. Initial temperature and volume of the gas are $T$ and $V$ respectively. If gas expand to $27V$ then its temperature will be comeView Solution
- 3A Carnot engine whose sink is at $300\, K$ has an efficiency of $40\%.$ By how much should the temperature of source be increased so as to increase its efficiency by $50\%$ of original efficiency ..... $K$View Solution
- 4The volume of air increases by $5\%$ in its adiabatic expansion. The percentage decrease in its pressure will be ...... $\%$View Solution
- 5Two moles of helium gas are taken over the cycle $ABCDA$, as shown in the $P-T$ diagram. Assuming the gas to be ideal the work done on the gas in taking it from $A $ to $B$ is ...... $R$View Solution

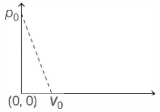
- 6One mole of ideal gas undergoes a linear process as shown in the figure below. Its temperature expressed as a function of volume $V$ isView Solution
- 7When $1\, kg$ of ice at $0^o C$ melts to water at $0^o C,$ the resulting change in its entropy, taking latent heat of ice to be $80\, cal/gm,$ is ...... $cal/K$View Solution
- 8View SolutionEntropy of a system decreases .........
- 9View SolutionIn a reversible isochoric change
- 10A tyre filled with air $({27^o}C,$ and $2$ atm) bursts, then what is temperature of air ....... $^oC$ $(\gamma = 1.5)$View Solution
