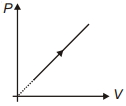
Suppose ideal gas equation follows $V{P^3}$= constant. Initial temperature and volume of the gas are $T$ and $V$ respectively. If gas expand to $27V$ then its temperature will be come
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Even Carnot engine cannot give $100\%$ efficiency because we cannotView Solution
- 2A Carnot's heat engine works between the temperatures $427^{\circ} C$ and $27^{\circ} C$. $...........\,kcal / s$ amount of heat should it consume per second to deliver mechanical work at the rate of $1.0\,kW$View Solution
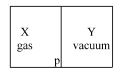
- 3A closed container is fully insulated from outside. One half of it is filled with an ideal gas $X$ separated by a plate Pfrom the other half $Y$ which contains a vacuum as shown in figure. When $P$ is removed, $X$ moves into $Y$. Which of the following statements is correct?View Solution
- 4$P_i, V_i$ and $P_f$, $V_f$ are initial and final pressures and volumes of a gas in a thermodynamic process respectively. If $PV^n =$ constant, then the amount of work done isView Solution
- 5View SolutionCompressed air in the tube of a wheel of a cycle at normal temperature suddenly starts coming out from a puncture. The air inside
- 6$540$ calories of heat converts $1$ cubic centimeter of water at $100\,^oC$ into $1671$ cubic centimeter of steam at $100\,^oC$ at a pressure of one atmosphere. Then the work done against the atmospheric pressure is nearly .......... $cal$View Solution
- 7View SolutionTwo gases are said to be in thermal equilibrium when they have same
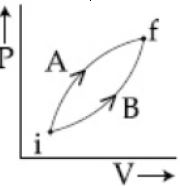
- 8Following figure shows two processes $A$ and $B$ for a gas. If $\Delta Q_A$ and $\Delta Q_B$ are the amount of heat absorbed by the system in two cases, and $\Delta U_A$ and $\Delta U_B$ are changes in internal energies, respectively, thenView Solution
- 9View SolutionBy opening the door of a refrigerator placed inside a room you
- 10View SolutionDuring the thermodynamic process shown in figure for an ideal gas