$(A)$ If $V_2=2 V_1$ and $T_2=3 T_1$, then the energy stored in the spring is $\frac{1}{4} P_1 V_1$
$(B)$ If $V_2=2 V_1$ and $T_2=3 T_1$, then the change in internal energy is $3 P_1 V_1$
$(C)$ If $V_2=3 V_1$ and $T_2=4 T_1$, then the work done by the gas is $\frac{7}{3} P_1 V_1$
$(D)$ If $V_2=3 V_1$ and $T_2=4 T_1$, then the heat supplied to the gas is $\frac{17}{6} P_1 V_1$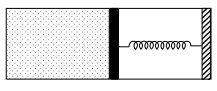
$W =\int PdV = P _1\left( V _2- V _1\right)+\frac{ kx ^2}{2}= P _1\left( V _2- V _1\right)+\frac{\left( P _2- P _1\right)\left( V _2- V _1\right)}{2}$
$\Delta U = nC _{ V } \Delta T =\frac{3}{2}\left( P _2 V _2- P _1 V _1\right)$
$Q = W +\Delta U$
$\text { Case I: } \Delta U =3 P _1 V _1, W =\frac{5 P _1 V _1}{4}, Q =\frac{17 P _1 V _1}{4}, U _{\text {spring }}=\frac{ P _1 V _1}{4} $
$\text { Case II: } \Delta U =\frac{9 P _1 V _1}{2}, W =\frac{7 P _1 V _1}{3}, Q =\frac{41 P _1 V _1}{6}, U _{\text {spring }}=\frac{ P _1 V _1}{3}$
Download our appand get started for free
Similar Questions
- 1During an experiment, an ideal gas is found to obey a condition $\frac{{{P^2}}}{\rho }$ = constant [$\rho =$ density of the gas]. The gas is initially at temperature $T,$ pressure $P$ and density $\rho$ . The gas expands such that density changes to $\rho/2$View Solution
- 2A cyclic process of a thermodynamic system is taken through $a$ $b$ $c$ $d$ $a$. The work done by the gas along the path $b$ $c$ isView Solution
- 3A cyclic process $ABCD$ is shown in the $p-V$ diagram. Which of the following curves represents the same process if $BC \& DA$ are isothermal processesView Solution
- 4In the diagrams $(i)$ to $(iv)$ of variation of volume with changing pressure is shown. A gas is taken along the path $ABCD. $ The change in internal energy of the gas will beView Solution
- 5A vertical cylinder with heat-conducting walls is closed at the bottom and is fitted with a smooth light piston. It contains one mole of an ideal gas. The temperature of the gas is always equal to the surrounding’s temperature, $T_0$. The piston is moved up slowly to increase the volume of the gas to $\eta$ times. Which of the following is incorrect?View Solution
- 6View SolutionNeon gas of a given mass expands isothermally to double volume. What should be the further fractional decrease in pressure, so that the gas when adiabatically compressed from that state, reaches the original state?
- 7A certain mass of gas at $273 K$ is expanded to $81$ times its volume under adiabatic condition. If $\gamma = 1.25$ for the gas, then its final temperature is ..... $^oC$View Solution
- 8View SolutionAn enclosed ideal gas is taken through a cycle as shown in the figure. Then
- 9A Carnot engine operates between ${227^o}C$ and ${27^o}C.$ Efficiency of the engine will beView Solution
- 10A Carnot's engine working between $400\, K$ and $800\, K$ has a work output of $1200\, J$ per cycle. The amount of heat energy supplied to the engine from the source in each cycle is ........... $J$View Solution
