In the diagrams $(i)$ to $(iv)$ of variation of volume with changing pressure is shown. A gas is taken along the path $ABCD. $ The change in internal energy of the gas will be
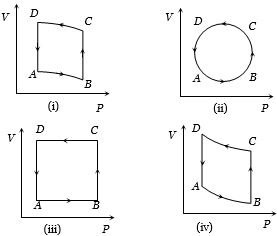

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Suppose ideal gas equation follows $V{P^3}$= constant. Initial temperature and volume of the gas are $T$ and $V$ respectively. If gas expand to $27V$ then its temperature will be comeView Solution
- 2$1c{m^3}$ of water at its boiling point absorbs $540$ calories of heat to become steam with a volume of $1671c{m^3}$.If the atmospheric pressure = $1.013 \times {10^5}N/{m^2}$ and the mechanical equivalent of heat = $4.19J/calorie$, the energy spent in this process in overcoming intermolecular forces is ..... $cal$View Solution
- 3View SolutionA container that suits the occurrence of an isothermal process should be made of
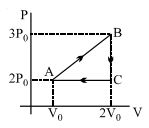
- 4View SolutionIn the above thermodynamic process, the correct statement is
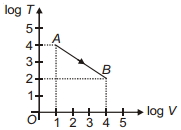
- 5Figure shows, the adiabatic curve on a $\log T$ and log $V$ scale performed on ideal gas. The gas is ............View Solution
- 6In case of an adiabatic process the correct relation in terms of pressure $p$ and density $\rho $ of a gas isView Solution
- 7An ideal gas follows a process $PT =$ constant. The correct graph between pressure $\&$ volume isView Solution
- 8The graph of pressure $(P)$ and volume $(V)$ according to $PV^n = C$, here $n = 1.4$View Solution
- 9View SolutionTwo gases have the same initial pressure, volume and temperatue. They expand to the same final volume, one adiabatically and the other isothermally, if the two gases are compressed to the same final volume
- 10A monoatomic gas is taken through a process $TP^{-1/3} =$ constant. If heat is given to the gasView Solution
