A carnot engine has the same efficiency between $800 K$ to $500 K$ and $x\, K$ to $600 K.$ The value of $x$ is ...... $K$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Find the change in the entropy in the following process $100 \,gm$ of ice at $0°C$ melts when dropped in a bucket of water at $50°C$ (Assume temperature of water does not change) ..... $ cal/K$View Solution
- 2View SolutionA perfect gas contained in a cylinder is kept in vacuum. If the cylinder suddenly bursts, then the temperature of the gas
- 3If $\gamma $ denotes the ratio of two specific heats of a gas, the ratio of slopes of adiabatic and isothermal $PV$ curves at their point of intersection isView Solution
- 4Heat is given to an ideal gas in an isothermal process.View Solution
$A.$ Internal energy of the gas will decrease.
$B.$ Internal energy of the gas will increase.
$C.$ Internal energy of the gas will not change.
$D.$ The gas will do positive work.
$E.$ The gas will do negative work.
Choose the correct answer from the options given below :
- 5The temperature of $3.00\, {mol}$ of an ideal diatomic gas is increased by $40.0^{\circ} {C}$ without changing the pressure of the gas. The molecules in the gas rotate but do not oscillate. If the ratio of change in internal energy of the gas to the amount of workdone by the gas is $\frac{{x}}{10} .$ Then the value of ${x}$ (round off to the nearest integer) is ..... . $\left(\right.$ Given $\left.{R}=8.31\, {J} {mol}^{-1} {K}^{-1}\right)$View Solution
- 6An engine is supposed to operate between two reservoirs at temperature $727°C$ and $227°C.$ The maximum possible efficiency of such an engine isView Solution
- 7Air is filled in a motor tube at ${27^o}C$ and at a pressure of $8$ atmospheres. The tube suddenly bursts, then temperature of air is $[{\rm{Given}}\,\,\gamma \,{\rm{of}}\,{\rm{air}} = \,1.5]$View Solution
- 8$n$ moles of a van der Waals' gas obeying the equation of state $\left(p+\frac{n^2 a}{V^2}\right)(V-n b)=n R T$, where $a$ and $b$ are gas dependent constants, is made to undergo a cyclic process that is depicted by a rectangle in the $p-V$ diagram as shown below. What is the heat absorbed by the gas in one cycle?View Solution
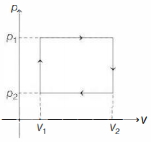
- 9The $P-V$ graph of an ideal gas cycle is shown here as below. The adiabatic process is described byView Solution
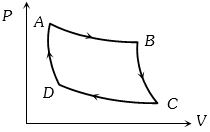
- 10$Assertion :$ In free expansion of an ideal gas, the entropy increases.View Solution
$Reason :$ Entropy increases in all natural processes.
