Download our appand get started for free
Similar Questions
- 1View SolutionWhen you make ice cubes, the entropy of water
- 2One mole of an ideal monoatomic gas undergoes the following four reversible processes:View Solution
Step $1$ It is first compressed adiabatically from volume $8.0 \,m ^{3}$ to $1.0 \,m ^{3}$.
Step $2$ Then expanded isothermally at temperature $T_{1}$ to volume $10.0 \,m ^{3}$.
Step $3$ Then expanded adiabatically to volume $80.0 \,m ^{3}$.
Step $4$ Then compressed isothermally at temperature $T_{2}$ to volume $8.0 \,m ^{3}$.
Then, $T_{1} / T_{2}$ is
- 3View SolutionIn a thermodynamic system working substance is ideal gas, its internal energy is in the form of
- 4An ideal heat engine exhausting heat at $77\,^oC$. To have a $30\%$ efficiency. It must take heat at...... $^oC$View Solution
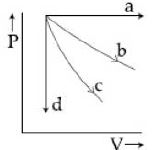
- 5View SolutionThe given diagram shows four processes i.e., isochoric, isobaric, isothermal and adiabatic. The correct assignment of the processes, in the same order is given by
- 6One mole of ${O_2}$ gas having a volume equal to $22.4$ litres at ${0^o}C$ and $1$ atmospheric pressure in compressed isothermally so that its volume reduces to $11.2$ litres. The work done in this process is ...... $J$View Solution
- 7A gas is compressed from a volume of $2\,m^3$ to a volume of $1\, m^3$ at a constant pressure of $100\, N/m^2$. Then it is heated at constant volume by supplying $150\, J$ of energy. As a result, the internal energy of the gasView Solution
- 8$1c{m^3}$ of water at its boiling point absorbs $540$ calories of heat to become steam with a volume of $1671c{m^3}$.If the atmospheric pressure = $1.013 \times {10^5}N/{m^2}$ and the mechanical equivalent of heat = $4.19J/calorie$, the energy spent in this process in overcoming intermolecular forces is ..... $cal$View Solution
- 9Answer the following by appropriately matching the lists based on the information given in the paragraph.View Solution
In a thermodynamics process on an ideal monatomic gas, the infinitesimal heat absorbed by the gas is given by $T \Delta X$, where $T$ is temperature of the system and $\Delta X$ is the infinitesimal change in a thermodynamic quantity $X$ of the system. For a mole of monatomic ideal gas
$X=\frac{3}{2} R \ln \left(\frac{T}{T_A}\right)+R \ln \left(\frac{V}{V_A}\right)$. Here, $R$ is gas constant, $V$ is volume of gas, $T_A$ and $V_A$ are constants.
The $List-I$ below gives some quantities involved in a process and $List-II$ gives some possible values of these quantities.
List-$I$ List-$II$ $(I)$ Work done by the system in process $1 \rightarrow 2 \rightarrow 3$ $(P)$ $\frac{1}{3} R T_0 \ln 2$ $(II)$ Change in internal energy in process $1 \rightarrow 2 \rightarrow 3$ $(Q)$ $\frac{1}{3} RT _0$ $(III)$ Heat absorbed by the system in process $1 \rightarrow 2 \rightarrow 3$ $(R)$ $R T _0$ $(IV)$ Heat absorbed by the system in process $1 \rightarrow 2$ $(S)$ $\frac{4}{3} RT _0$ $(T)$ $\frac{1}{3} RT _0(3+\ln 2)$ $(U)$ $\frac{5}{6} RT _0$ If the process carried out on one mole of monatomic ideal gas is as shown in figure in the PV-diagram with $P _0 V _0=\frac{1}{3} RT _0$, the correct match is,
$(1)$$I \rightarrow Q, II \rightarrow R , III \rightarrow P , IV \rightarrow U$
$(2)$ $I \rightarrow S , II \rightarrow R , III \rightarrow Q , IV \rightarrow T$
$(3)$ $I \rightarrow Q , II \rightarrow R , III \rightarrow S , IV \rightarrow U$
$(4)$ $I \rightarrow Q , II \rightarrow S , III \rightarrow R , IV \rightarrow U$
($2$) If the process on one mole of monatomic ideal gas is an shown is as shown in the $TV$-diagram with $P _0 V _0=\frac{1}{3} RT _0$, the correct match is
$(1)$ $I \rightarrow S, II \rightarrow T, III \rightarrow Q , IV \rightarrow U$
$(2)$ $I \rightarrow P , II \rightarrow R, III \rightarrow T , IV \rightarrow S$
$(3)$ $I \rightarrow P, II \rightarrow, III \rightarrow Q, IV \rightarrow T$
$(4)$ $I \rightarrow P, II \rightarrow R, III \rightarrow T, IV \rightarrow P$
Give the answer or quetion $(1)$ and $(2)$
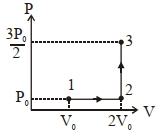
- 10An ideal gas at pressure $P$ and volume $V$ is expanded to volume$ 2V.$ Column $I$ represents the thermodynamic processes used during expansion. Column $II$ represents the work during these processes in the random order.:View Solution
Column $I$ Column $II$ $(p)$ isobaric $(x)$ $\frac{{PV(1 - {2^{1 - \gamma }})}}{{\gamma - 1}}$ $(q)$ isothermal $(y)$ $PV$ $(r)$ adiabatic (z) $PV\,\iota n\,2$ The correct matching of column $I$ and column $II$ is given by
