| Column $I$ | Column $II$ |
| $(p)$ isobaric | $(x)$ $\frac{{PV(1 - {2^{1 - \gamma }})}}{{\gamma - 1}}$ |
| $(q)$ isothermal | $(y)$ $PV$ |
| $(r)$ adiabatic | (z) $PV\,\iota n\,2$ |
The correct matching of column $I$ and column $II$ is given by
$P_{i}=P$
$(P) \rightarrow(y)$ isobaric process
$\mathrm{W}=\mathrm{P} \Delta \mathrm{V}=\mathrm{PV}$
$(q) \rightarrow(z)$ isothermal
$\mathrm{W}=\mathrm{nRT} \ln \frac{\mathrm{V}_{\mathrm{f}}}{\mathrm{V}_{\mathrm{i}}}=\mathrm{PV} \ln 2$
$(r) \rightarrow(x)$ Adiabatic
$P_{f}=\left(\frac{V_{i}}{V_{f}}\right)^{\gamma} P_{i}=2^{\gamma} P$
$W=\frac{P_{f} V_{f}-P_{i} V_{i}}{1-\gamma}=\frac{\left(2^{-\gamma} P\right)(2 V)-P V}{1-\gamma}$
$\Rightarrow w=\frac{P V\left(1-2^{1-\gamma}\right)}{\gamma-1}$
Download our appand get started for free
Similar Questions
- 1When heat energy of $1500\; Joules$, is supplied to a gas at constant pressure $2.1 \times {10^5}\;N/{m^2}$, there was an increase in its volume equal to $2.5 \times {10^{ - 3}}\;{m^3}$. The increase in internal energy of the gas in Joules is ...... $J$View Solution
- 2A gas can be taken from $A$ to $B$ via two different processes $ACB$ and $ADB$. When path $ACB$ is used $60\, J$ of heat flows into the system and $30\, J$ of work is done by the system. If path $ADB$ is used work down by the system is $10\, J$. the heat flow into the system in path $ADB$ is ..... $J$View Solution
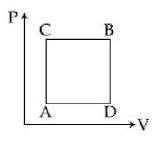
- 3An ideal monoatomic gas is confined in a horizontal cylinder by a spring loaded piston (as shown in the figure). Initially the gas is at temperature $T _1$, pressure $P_1$ and volume $V_1$ and the spring is in its relaxed state. The gas is then heated very slowly to temperature $T_2$, pressure $P _2$ and volume $V _2$. During this process the piston moves out by a distance $x$. Ignoring the friction between the piston and the cylinder, the correct statement$(s)$ is(are)View Solution
$(A)$ If $V_2=2 V_1$ and $T_2=3 T_1$, then the energy stored in the spring is $\frac{1}{4} P_1 V_1$
$(B)$ If $V_2=2 V_1$ and $T_2=3 T_1$, then the change in internal energy is $3 P_1 V_1$
$(C)$ If $V_2=3 V_1$ and $T_2=4 T_1$, then the work done by the gas is $\frac{7}{3} P_1 V_1$
$(D)$ If $V_2=3 V_1$ and $T_2=4 T_1$, then the heat supplied to the gas is $\frac{17}{6} P_1 V_1$
- 4In an Isothermal change, the change in pressure and volume of a gas can be represented for three different temperature; $T _3 > T _2 > T _1$ as :View Solution
- 5The coefficient of performance of a refrigerator is $5$ . If the temperature inside freezer is $-20\,^oC$ , the temperature of the surroundings to which it rejects heat is ....... $^oC$View Solution
- 6A motor-car tyre has a pressure of $2\, atm$ at $27\,^oC$. It suddenly burst's. If $\left( {\frac{{{C_p}}}{{{C_v}}}} \right) = 1.4$ for air, find the resulting temperatures (Given $4^{1/7} = 1.219$)View Solution
- 7One mole of a monatomic ideal gas is taken through a cycle $ABCDA$ as shown in the $P-V$ diagram. Column $II$ gives the characteristics involved in the cycle. Match them with each of the processes qiven in Column $I$View Solution
Column $I$ Column $II$ $(A)$ Process $A \rightarrow B$ $(p)$ Internal energy decreases. $(B)$ Process $B \rightarrow C$ $(q)$ Internal energy increases. $(C)$ Process $C \rightarrow D$ $(r)$ Heat is lost. $(D)$ Process $D \rightarrow A$ $(s)$ Heat is gained. $(t)$ Work is done on the gas. 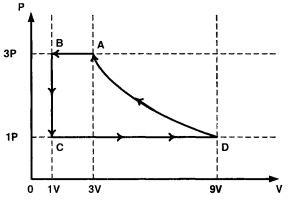
- 8Consider one mole of helium gas enclosed in a container at initial pressure $P_1$ and volume $V_1$. It expands isothermally to volume $4 V_1$. After this, the gas expands adiabatically and its volume becomes $32 V_1$. The work done by the gas during isothermal and adiabatic expansion processes are $W_{\text {iso }}$ and $W_{\text {adia, }}$ respectively. If the ratio $\frac{W_{\text {iso }}}{W_{\text {adia }}}=f \ln 2$, then $f$ is. . . . . . . .View Solution
- 9A mono atomic gas is supplied the heat $Q$ very slowly keeping the pressure constant. The work done by the gas will beView Solution
- 10In the following figure, four curves $A, B, C$ and $D$ are shown. The curves areView Solution