If $\Delta U$ and $\Delta W$ represent the increase in internal energy and work done by the system respectively in a thermodynamical process, which of the following is true?
AIPMT 1998,AIPMT 2010, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
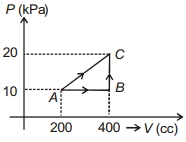
- 1If a gas is taken from $A$ to $C$ through $B$ then heat absorbed by the gas is $8 \,J$. Heat absorbed by the gas in taking it from $A$ to $C$ directly is ............. $J$View Solution
- 2$300 \,cal$. of heat is given to a heat engine and it rejects $225 \,cal$. of heat. If source temperature is $227^{\circ} C$, then the temperature of sink will be____${ }^{\circ} C$.View Solution
- 3Avessel with open mouth contains air at $60^oC$. When the vessel is heated upto temperature $T$, one fourth of the air goes out. The value of $T$ is ..... $^oC$View Solution
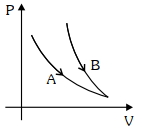
- 4Choose the correct statement for processes $A$ & $B$ shown in figure.View Solution
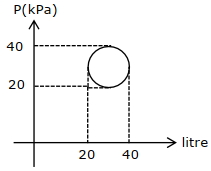
- 5In the reported figure, heat energy absorbed by a system in going through a cyclic process is $......\,\pi J$View Solution
- 6View SolutionAn ideal diatomic gas is heated at constant pressure. The ratio of the work done to the heat supplied is
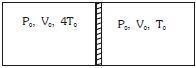
- 7Figure shows a cylindrical adiabatic container of total volume $2V_0$ divided into two equal parts by a conducting piston (which is free to move). Each part containing identical gas at pressure $P_0$ . Initially temperature of left and right part is $4T_0$ and $T_0$ respectively. An external force is applied on the piston to keep the piston at rest. Find the value of external force required when thermal equilibrium is reached. ( $A =$ Area of piston)View Solution
- 8Choose the incorrect statement from the followingView Solution
$S1:$ The efficiency of a heat engine can be $1,$ but the coefficient of performance of a refrigerator can never be infinity.
$S2:$ The first law of thermodynamics is basically the principle of conservation of energy.
$S3:$ The second law of thermodynamics does not allow several phenomena consistent with the first law.
$S4:$ A process,whose only result is to transfer heat from a colder to a hotter object, is impossible.
- 9$Assertion :$ Reversible systems are difficult to find in real world.View Solution
$Reason :$ Most processes are dissipative in nature. - 10View SolutionA container that suits the occurrence of an isothermal process should be made of
