In a heat engine, the temperature of the source and sink are $500\, K$ and $375\, K$. If the engine consumes $25\times10^5\, J$ per cycle, the work done per cycle is
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An ideal gas expands isothermally from a volume ${V_1}$ to ${V_2}$ and then compressed to original volume ${V_1}$adiabatically. Initial pressure is ${P_1}$ and final pressure is ${P_3}$. The total work done is $W$. ThenView Solution
- 2Given below are two statements : one is labelled as Assertion A and the other is labelled as Reason $R$. Assertion A : Efficiency of a reversible heat engine will be highest at $-273^{\circ} C$ temperature of cold reservoir.View Solution
Reason $R$ : The efficiency of Carnot's engine depends not only on temperature of cold reservoir but it depends on the temperature of hot reservoir too and is given as $\eta=\left(1-\frac{ T _2}{ T _1}\right)$.
In the light of the above statements, choose the correct answer from the options given below
- 3The volume of an ideal gas $(\gamma=1.5)$ is changed adiabatically from $5$ litres to $4$ litres. The ratio of initial pressure to final pressure is:View Solution
- 4Initial pressure and volume of a gas are $P$ and $V$ respectively. First it is expanded isothermally to volume $4V$ and then compressed adiabatically to volume $V$ . The final pressure of gas will be (given $\gamma = 3/2$ )View Solution
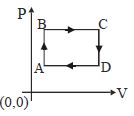
- 5The figure shows $P-V$ diagram of a thermodynamic cycle. Which corresponding curve is correct?View Solution
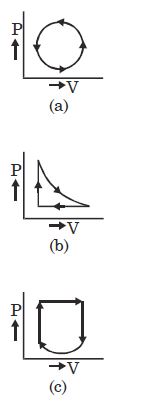
- 6View SolutionWhat is the nature of change in internal energy in the following three thermodynamical processes shown in figure
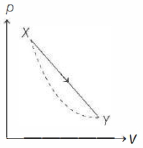
- 7In the $p-V$ diagram below, the dashed curved line is an adiabat.For a process that is described by a straight line joining two points $X$ and $Y$ on the adiabat (solid line in the diagram) heat is (Hint consider the variation in temperature from $X$ to $Y$ along the straight line)View Solution
- 8View SolutionIn an isothermal change, an ideal gas obeys
- 9In a heat engine, the temperature of the source and sink are $500\, K$ and $375\, K$. If the engine consumes $25\times10^5\, J$ per cycle, the work done per cycle isView Solution
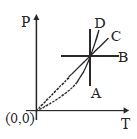
- 10Following figure shows $P-T$ graph for four processes $A, B, C$ and $D$. Select the correct alternativeView Solution