One mole of a monatomic ideal gas is taken along two cyclic processes $E \rightarrow F \rightarrow G \rightarrow E$ and $E \rightarrow F \rightarrow H \rightarrow$ E as shown in the $PV$ diagram. The processes involved are purely isochoric, isobaric, isothermal or adiabatic. $Image$
Match the paths in List $I$ with the magnitudes of the work done in List $II$ and select the correct answer using the codes given below the lists.
| List $I$ | List $I$ |
| $P.$ $\quad G \rightarrow E$ | $1.$ $\quad 160 P_0 V_0 \ln 2$ |
| $Q.$ $\quad G \rightarrow H$ | $2.$ $\quad 36 P _0 V _0$ |
| $R.$ $\quad F \rightarrow H$ | $3.$ $\quad 24 P _0 V _0$ |
| $S.$ $\quad F \rightarrow G$ | $4.$ $\quad 31 P_0 V_0$ |
Codes: $ \quad \quad P \quad Q \quad R \quad S $ 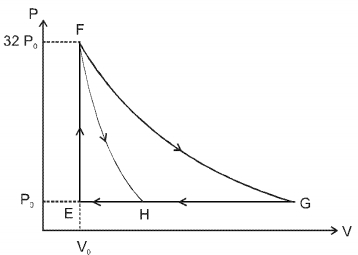
IIT 2013, Diffcult
In $F \rightarrow G$ work done in isothermal proces is $n R T \ln \left(\frac{V_f}{V_i}\right)=32 P_0 V_0 \ln \left(\frac{32 V_0}{V_0}\right)$
$=32 P_0 V_0 \ln 2^5=160 P_0 V_0 \ln 2 $
$\ln G \rightarrow E, \Delta W=P_0 \Delta V=P_0\left(31 V_0\right)=31 P_0 V_0$
In $G \rightarrow H$ work done is less than $31 P_0 V_0$ i.e., $24 P_0 V_0$
In $F \rightarrow H$ work done is $36 P _0 V _0$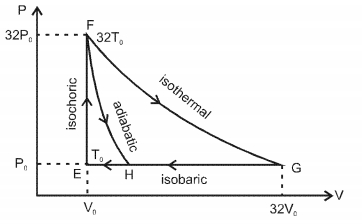
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
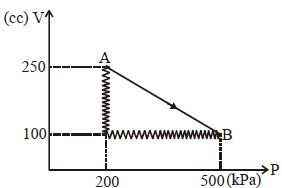
- 1A monoatomic gas is taken along path $AB$ as shown. Calculate change in internal energy of systemView Solution
- 2View SolutionThe adiabatic Bulk modulus of a diatomic gas at atmospheric pressure is
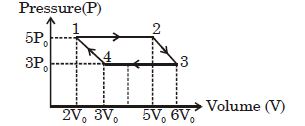
- 3An ideal monoatomic gas is taken round the cycle $12341$ shown in the figure ( $p\,-\,V$ diagram). The work done in one cycle is ?View Solution
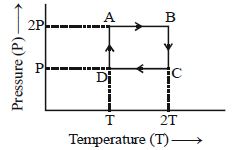
- 4One mole of an ideal gas having initial volume $V$, pressure $2P$ and temperature $T$ undergoes a cyclic process $ABCDA$ as shown below : The net work done in the complete cycle isView Solution
- 5An engine takes in $5$ moles of air at $20\,^{\circ} C$ and $1$ $atm,$ and compresses it adiabaticaly to $1 / 10^{\text {th }}$ of the original volume. Assuming air to be a diatomic ideal gas made up of rigid molecules, the change in its internal energy during this process comes out to be $X\, kJ$. The value of $X$ to the nearest integer isView Solution
- 6For an adiabatic expansion of an ideal gas, the fractional change in its pressure is equal to (where $\gamma$ is the ratio of specific heats):View Solution
- 7$Assertion :$ Reversible systems are difficult to find in real world.View Solution
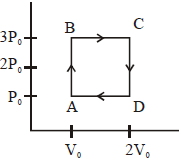
$Reason :$ Most processes are dissipative in nature. - 8An engine operates by taking a monatomic ideal gas through the cycle shown in the figure. The percentage efficiency of the engine is close to $.......\%$View Solution
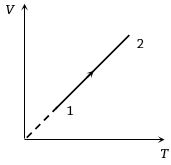
- 9Volume versus temperature graph of two moles of helium gas is as shown in figure. The ratio of heat absorbed and the work done by the gas in process $1-2$ isView Solution
- 10View SolutionWhich relation is correct for isometric process
