In kinetic theory of gases, a molecule of mass m of an ideal gas collides with a wall of vessel with velocity $V.$ The change in the linear momentum of the molecule is
AIIMS 1997, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionBoyle's law holds for an ideal gas during
- 2View SolutionWhich of the following statement is true
- 3If the collision frequency of hydrogen molecules in a closed chamber at $27^{\circ} \mathrm{C}$ is $\mathrm{Z}$, then the collision frequency of the same system at $127^{\circ} \mathrm{C}$ is :View Solution
- 4A thermally insulated vessel contains an ideal gas of molecular mass $M$ and ratio of specific heats $\gamma$. It is moving with speed $v$ and it's suddenly brought to rest. Assuming no heat is lost to the surroundings, its temperature increases byView Solution
- 5A given sample of an ideal gas occupies a volume $V$ at a pressure $P$ and absolute temperature $T.$ The mass of each molecule of the gas is $m.$ Which of the following gives the density of the gas $?$View Solution
- 6At $27°C$ temperature, the kinetic energy of an ideal gas is ${E_1}$. If the temperature is increased to $327°C,$ then kinetic energy would beView Solution
- 7At temperature $T,$ the $r.m.s.$ speed of helium molecules is the same as $r.m.s.$ speed of hydrogen molecules at normal temperature and pressure. The value of $T$ is ....... $^oC$View Solution
- 8$PV$ versus $T$ graph of equal masses of ${H_2}$, $He$ and ${O_2}$ is shown in fig. Choose the correct alternativeView Solution
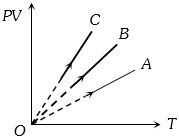
- 9The temperature of an ideal gas at atmospheric pressure is $300\,K$ and volume $1\,m^3$. If temperature and volume become double, then pressure will beView Solution
- 10Ideal monoatomic gas is taken through a process $dQ = 2dU.$ The molar heat capacity for the processView Solution
