The temperature of an ideal gas at atmospheric pressure is $300\,K$ and volume $1\,m^3$. If temperature and volume become double, then pressure will be
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1At what temperature volume of an ideal gas at $0°C$ becomes triple ....... $^oC$View Solution
- 2Which of the following shows the correct relationship between the pressure $'P'$ and density $\rho $ of an ideal gas at constant temperature ?View Solution
- 3If the pressure of an ideal gas contained in a closed vessel is increased by $0.5\%,$ the increase in temperature is $2K.$ The initial temperature of the gas is ...... $^oC$View Solution
- 4An ideal gas is enclosed in a container of volume $V$ at a pressure $P$. It is being pumped out of the container by using a pump with stroke volume $v$. What is final pressure in container after $n$-stroke of the pump? (assume temperature remains same)View Solution
- 5The specific heat of the mixture of two gases at constant volume is $\frac {13}{6}\,R$ . The ratio of number of moles of first gas to second is $1 : 2$. The respective gases may beView Solution
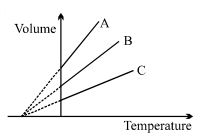
- 6The expansion of an ideal gas of mass $m$ at a constant pressure $P$ is given by the straight line $B$. Then the expansion of the same ideal gas of mass $2 \,\,m$ at a pressure $2P$ is given by the straight lineView Solution
- 7View SolutionThe relation between two specific heats of a gas is
- 8The molar heat capacities of a mixture of two gases at constant volume is $13 R / 6$. The ratio of number of moles of the first gas to the second is $1: 2$. The respective gases may beView Solution
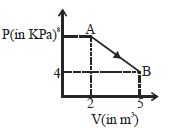
- 9A mole of an ideal diatomic gas undergoes a transition from $A$ to $B$ along a path $AB$ as shown in the figure, The change in internal energy of the gas during the transition is ...... $kJ$View Solution
- 10View SolutionVariation of atmospheric pressure, with height from earth is ................
