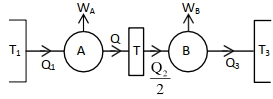
Two carnot engines $A$ and $B$ operate in series such that engine $A$ absorbs heat at $T_{1}$ and rejects heat to a sink at temperature $T$. Engine $B$ absorbs half of the heat rejected by engine $A$ and rejects heat to the sink at ${T}_{3}$. When workdone in both the cases is equal, the value of ${T}$ is
JEE MAIN 2021, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionEntropy of a system decreases .........
- 2$Assertion :$ The isothermal curves intersect each other at a certain point.View Solution
$Reason :$ The isothermal change takes place slowly, so, the isothermal curves have very little slope. - 3Heat energy of $735\,J$ is given to a diatomic gas allowing the gas to expand at constant pressure. Each gas molecule rotates around an internal axis but do not oscillate. The increase in the internal energy of the gas will be $..........\,J$View Solution
- 4Two Carnot engines $A$ and $B$ are operated in succession. The first one, $A$ receives heat from a source at $T_1 = 800\, K$ and rejects to sink at $T_2K$. The second engine $B$ receives heat rejected by the first engine and rejects to another sink at $T_3 = 300\, K$. If the work outputs of two engines are equal, then the value of $T_2$ is ...... $K$View Solution
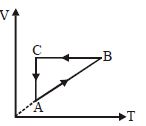
- 5A cyclic process $ABCA$ is shown in the $V-T$ diagram. Process on the $P-V$ diagram isView Solution
- 6The latent heat of vaporisation of water is $2240\, J/gm$. If the work done in the process of expansion of $1 \,g$ is $168 \,J$, then increase in internal energy is ....... $J$View Solution
- 7A gas expands under constant pressure $P$ from volume ${V_1}$ to ${V_2}$. The work done by the gas isView Solution
- 8If the temperature of sink is at absolute zero, then the efficiency of Carnot engine will be ........ $\%$View Solution
- 9A solid body of constant heat capacity $1\ J/^o C$ is being heated by keeping it in contact with reservoirs in two ways :View Solution
$(i)$ Sequentially keeping in contact with $2$ reservoirs such that each reservoir supplies same amount of heat.
$(ii)$ Sequentially keeping in contact with $8$ reservoirs such that each reservoir supplies same amount of heat.
In both the cases body is brought from initial temperature $100^o C$ to final temperature $200^o C$. Entropy change of the body in the two cases respectively is :
- 10An engineer claims to have made an engine delivering $10 kW$ power with fuel consumption of $1\,g\,{s^{ - 1}}$. The calorific value of fuel is $2k cal/g$. His claimView Solution