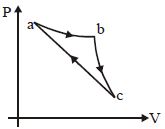
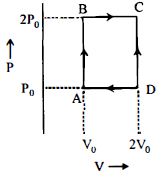
In the reported figure, there is a cyclic process $ABCDA$ on a sample of $1\, {mol}$ of a diatomic gas. The temperature of the gas during the process ${A} \rightarrow {B}$ and ${C} \rightarrow {D}$ are ${T}_{1}$ and ${T}_{2}\left({T}_{1}\,>\,{T}_{2}\right)$ respectively.
Choose the correct option out of the following for work done if processes $B C$ and $D A$ are adiabatic.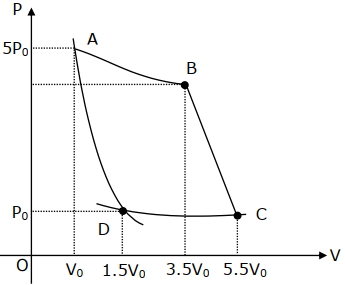
JEE MAIN 2021, Medium
Work done in adiabatic process $=\frac{-n R}{\gamma-1}\left(T_{f}-T_{i}\right)$
$\therefore W _{ AD }=\frac{- nR }{\gamma-1}\left( T _{2}- T _{1}\right)$
$\text { and } W _{ BC }=\frac{- nR }{\gamma-1}\left( T _{2}- T _{1}\right)$
$\therefore W _{ AD }= W _{ BC }$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An ideal gas at ${27^o}C$ is compressed adiabatically to $\frac{8}{{27}}$ of its original volume. If $\gamma = \frac{5}{3}$, then the rise in temperature is........ $K$View Solution
- 2$Q$ amount of heat is given to $0.5\ mole$ of an ide al mono-atomic gas by a process $TV^n$ constant. Following graph shows variation of temperature with $Q$ . Find value of $n$.View Solution
- 3During which of the following thermodynamic process represented by $P V$ diagram the heat energy absorbed by system may be equal to area under $P V$ graph?View Solution
- 4A perfect gas of a given mass is heated first in a small vessel and then in a large vessel, such that their volumes remain unchanged. The $P-T$ curves areView Solution
- 5Avessel with open mouth contains air at $60^oC$. When the vessel is heated upto temperature $T$, one fourth of the air goes out. The value of $T$ is ..... $^oC$View Solution
- 6An engine operates by taking $n\,moles$ of an ideal gas through the cycle $ABCDA$ shown in figure. The thermal efficiency of the engine is : (Take $C_v =1 .5\, R$, where $R$ is gas constant)View Solution
- 7A Carnot engine has an efficiency of $50 \%$ when its source is at a temperature $327^{\circ}\,C$. The temperature of the sink is $.........^{\circ} C$View Solution
- 8View SolutionWhich of the following is not a thermodynamics co-ordinate
- 9If a Carnot engine works between $127^{\circ} C$ and $527^{\circ} C$, then its efficiency is ...... $\%$View Solution
- 10In the $P-V$ diagram shown, the gas does $5\, J$ of work in isothermal process $ab$ and $4\,J$ in adiabatic process $bc$. .... $J$ will be the change in internal energy of the gas in straight path $c$ to $a$ ?View Solution