કાર્બએનાયન ની સ્થિરતા નો સાચો ક્રમ શોધો.
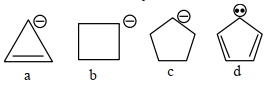

JEE MAIN 2024, Medium
d
As we know compound ($d$) is aromatic and the compound ($a$) is anti-aromatic. Hence compound
As we know compound ($d$) is aromatic and the compound ($a$) is anti-aromatic. Hence compound
($d$)is most stable and compound ($a$) is least stable among these in compound ($b$) and ($c$) carbon atom of that positive charge is $\mathrm{sp}^3$ hybridised they on the basis of angle strain theory compound ($c$) is more stable than compound ($b$).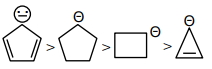
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View Solutionનીચે આપેલા માટે સૌથી વધુ સ્થાયી કાર્બોકેશાયન શોધી.
- 2View Solutionનીચેનામાંથી કયો પ્રબળ એસિડ છે?
- 3View Solutionનીચેની કયો સ્પીસીઝ સૌથી સ્થાયી છે?
- 4View Solutionઇલેક્ટ્રોન ચક્રિય વિસ્થાપન આગળ વધે છે એ
- 5View Solutionનીચેના કાર્બન કેટાયન ને ધ્યાનમાં લો. આ કાર્બોકેટાયનની સંલગ્ન સ્થિરતા કઈ છે?

- 6View Solutionઆપેલા પદાર્થોમાં એસિડિકતાનો સાચો ક્રમ કયો છે ?
- 7View Solutionઆલ્કીન સાથે બ્રોમિનની પ્રક્રિયા એ શેનું ઉદાહરણ છે ?
- 8${{\text{3}}^ \circ }{\text{ , }}{{\text{2}}^ \circ }{\text{ ,}}{{\text{1}}^ \circ }$ અને $\mathop C\limits^ \bullet {{\text{H}}_{\text{3}}}$ માં કાર્બન મૂલકોની સ્થિરતાનો ક્ર્મ ક્યો છેView Solution
- 9View Solutionનીચેના પૈકી કયું કાર્બોકેટાયન વધુ સ્થાયી છે ?
- 10નીચેનામાંથી કયા પરમાણુમાં શૂન્ય સિવાયના દ્વિધ્રુવી ચાકમાત્રા છે?View Solution
$(I)$ $1, 2$ - ડાયબ્રોમોઇથેન ની ગૌચ રચના
$(II)$ $1, 2$ -ડાયબ્રોમોઇથેન ની એન્ટિ રચના
$(III)$ ટ્રાન્સ - $1, 4$ -ડાયબ્રોમોસાયકલોહેકઝેન
$(IV)$ સિસ - $1, 4$ -ડાયબ્રોમોસાયકલોહેકઝેન
$(V)$ ટેટ્રાબ્રોમોઇથેન
$(VI)$ $1, 1$ - ડાયબ્રોમોસાયકલોહેકઝેન
