$n$ moles of a van der Waals' gas obeying the equation of state $\left(p+\frac{n^2 a}{V^2}\right)(V-n b)=n R T$, where $a$ and $b$ are gas dependent constants, is made to undergo a cyclic process that is depicted by a rectangle in the $p-V$ diagram as shown below. What is the heat absorbed by the gas in one cycle?
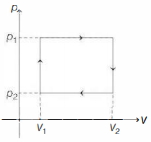

KVPY 2009, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionAn ideal gas, undergoing adiabatic change, has which of the following pressure temperature relationship?
- 2View SolutionThe state of an ideal gas was changed isobarically. The graph depicts three such isobaric lines. Which of the following is true about the pressures of the gas?
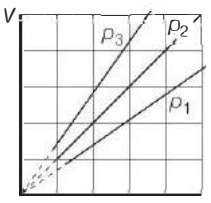
- 3View SolutionThe door of a working refrigerator is left open in a well insulated room. The temperature of air in the room will
- 4View SolutionWork done per mol in an isothermal change is
- 5If minimum possible work is done by a refrigerator in converting $100\; grams$ of water at $0^{\circ} C$ to ice, how much heat (in calories) is released to the surrounding at temperature $27^{\circ} C$ (Latent heat of ice $=80 Cal / gram$ ) to the nearest integer?View Solution
- 6View SolutionWhich of the following can not determine the state of a thermodynamic system
- 7An ideal heat engine working between temperature $T_1$ and $T_2 $ has an efficiency $\eta$, the new efficiency if both the source and sink temperature are doubled, will beView Solution
- 8A cyclic process $ABCD$ is shown in the figure $P-V$ diagram. Which of the following curves represent the same processView Solution
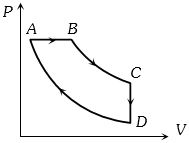
- 9Given below are two statementView Solution
Statement $-I$ : What $\mu$ amount of an ideal gas undergoes adiabatic change from state $\left( P _{1}, V _{1}, T _{1}\right)$ to state $\left( P _{2}, V _{2}, T _{2}\right)$, the work done is $W =\frac{1 R \left( T _{2}- T _{1}\right)}{1-\gamma}$, where $\gamma=\frac{ C _{ P }}{ C _{ V }}$ and $R =$ universal gas constant,
Statement $-II$ : In the above case. when work is done on the gas. the temperature of the gas would rise.
Choose the correct answer from the options given below
- 10View SolutionEntropy of a system decreases .........
