One mole of a diatomic ideal gas undergoes a cyclic process $ABC$ as shown in figure. The process $BC$ is adiabatic. The temperatures at $A, B$ and $C$ are $400\ K, 800\ K $ and $600\ K$ respectively. Choose the correct statement
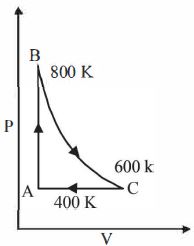

JEE MAIN 2014, Easy
In cyclic process, change in total internal energy is zero.
$\Delta {U_{cyclic}} = 0$
$\Delta {U_{BC}} = n{C_v}\Delta T = 1 \times \frac{{5R}}{2}\Delta T$
$Where,{C_v} = molar\,specific\,heat\,at\,constant\,volume.$
$For\,BC,\Delta T = - 200K$
$\therefore \,\,\Delta {U_{BC}} = - 500R$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1One mole of an ideal gas goes from an initial state $A$ to final state $B$ via two processes : It first undergoes isothermal expansion from volume $V$ to $3\, V$ and then its volume is reduced from $3\, V$ to $V$ at constant pressure. The correct $P-V$ diagram representing the two processes isView Solution
- 2Areversible adiabatic path on a $P-V$ diagram for an ideal gas passes through stateAwhere $P=0$.$7\times 10^5 \,\,N/ m^{-2}$ and $v = 0.0049 \,\,m^3$. The ratio of specific heat of the gas is $1.4$. The slope of path at $A$ is :View Solution
- 3Three processes form a thermodynamic cycle as shown on $P-V$ diagram for an ideal gas. Process $1 \rightarrow 2$ takes place at constant temperature $(300K$). Process $2 \rightarrow 3$ takes place at constant volume. During this process $40J$ of heat leaves the system. Process $3 \rightarrow 1$ is adiabatic and temperature $T_3$ is $275K$. Work done by the gas during the process $3 \rightarrow 1$ is ..... $J$View Solution
- 4View SolutionIn pressure-volume diagram given below, the isochoric, isothermal, and isobaric parts respectively, are
- 5A Carnot engine whose low temperature reservoir is at $7\,°C$ has an efficiency of $50\%$. It is desired to increase the efficiency to $70\%$. By how many degrees should the temperature of the high temperature reservoir be increased ....... $K$View Solution
- 6View SolutionThe temperature of an ideal gas is kept constant as it expands. The gas does external work. During this process, the internal energy of the gas
- 7A fixed amount of a gas undergoes a thermodynamic process as shown such that heat interaction along path $B \to C \to A$ is equal to the work done by the gas along path $A \to B \to C$. Then process $A \to B$ is :-View Solution
- 8An ideal gas undergoes change in its state from the initial state $I$ to the final state $F$ via two possible paths as shown below. Then,View Solution
- 9The temperature of $3.00\, {mol}$ of an ideal diatomic gas is increased by $40.0^{\circ} {C}$ without changing the pressure of the gas. The molecules in the gas rotate but do not oscillate. If the ratio of change in internal energy of the gas to the amount of workdone by the gas is $\frac{{x}}{10} .$ Then the value of ${x}$ (round off to the nearest integer) is ..... . $\left(\right.$ Given $\left.{R}=8.31\, {J} {mol}^{-1} {K}^{-1}\right)$View Solution
- 10View SolutionThe door of a working refrigerator is left open in a well insulated room. The temperature of air in the room will
