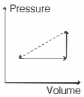
An ideal gas is made to undergo the cyclic process shown in the figure below. Let $\Delta W$ depict the work done, $\Delta U$ be the change in internal energy of the gas and $Q$ be the heat added to the gas. Sign of each of these three quantities for the whole cycle will be (0 refers to no change)

KVPY 2018, Medium
$(a)$ Given cyclic process is
Area under compression process $C A$ is more than area under expansion process AB. So, net work done is negative.
i.e. $\Delta W < 0$
Also, in a cyclic process, change in internal energy is zero.
i.e. $\Delta U=0$
Now, by using first law of thermodynamics, we have
$\Delta Q=0+\Delta W$
we see that, $\quad \Delta Q < 0$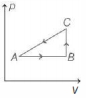
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1How much work to be done in decreasing the volume of and ideal gas by an amount of $2.4 \times {10^{ - 4}}{m^3}$ at normal temperature and constant normal pressure of .......$joule$ $1 \times {10^5}N/{m^2}$View Solution
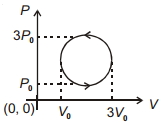
- 2View SolutionWork done in the cyclic process shown in figure is ...........
- 3When a system is taken from state $i$ to state $f$ along the path $iaf$, it is found that $Q=50$ $cal$ and $W=20$ $cal$ Along the path $ibf\ Q = 36\ cal. \ W$ along the path $ibf$ is ....... $ cal$View Solution
- 4A sample of gas expands from volume ${V_1}$ to ${V_2}$. The amount of work done by the gas is greatest when the expansion isView Solution
- 5View SolutionThe ratio of work done by an ideal monoatomic gas to the heat supplied to it in an isobaric process is
- 6A sample of gas at temperature $T$ is adiabatically expanded to double its volume. The work done by the gas in the process is $\left(\right.$ given, $\left.\gamma=\frac{3}{2}\right)$ :View Solution
- 7$5.6$ $liter$ of helium gas at $STP$ is adiabatically compressed to $0.7$ $liter$. Taking the initial temperature to be $T_1$, the work done in the process isView Solution
- 8View SolutionEntropy of a thermodynamic system does not change when this system is used for
- 9Match List $I$ with List $II$ :View Solution
List $I$ List $II$ $A$ Isothermal Process $I$ Work done by the gas decreases internal energy $B$ Adiabatic Process $II$ No change in internal energy $C$ Isochoric Process $III$ The heat absorbed goes partly to increase internal energy and partly to do work $D$ Isobaric Process $IV$ No work is done on or by the gas Choose the correct answer from the options given below :
- 10View SolutionAn adiabatic process occurs at constant
