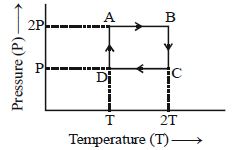
At constant pressure,
$\Delta \mathrm{W}=\mathrm{P}\left(\mathrm{V}_{\mathrm{t}}-\mathrm{V}_{\mathrm{i}}\right)=\mathrm{nR}\left(\mathrm{T}_{\mathrm{f}}-\mathrm{T}_{\mathrm{i}}\right)$
At constant temperature,
$\Delta \mathrm{W}=\mathrm{nRT} \ln \left(\frac{\mathrm{V}_{\mathrm{f}}}{\mathrm{V}_{\mathrm{i}}}\right)=\mathrm{nRT} \ln \left(\frac{\mathrm{P}_{\mathrm{i}}}{\mathrm{V}_{\mathrm{f}}}\right)$
Therefore, work done for path $\mathrm{AB}, \mathrm{BC}, \mathrm{CD}$ and
$DA$ respectively will be.
$\Delta \mathrm{W}_{\mathrm{AB}}=1 \times \mathrm{R} \times(2 \mathrm{T}-\mathrm{T})=\mathrm{RT}$
$\Delta \mathrm{W}_{\mathrm{BC}}=1 \times \mathrm{R} \times 2 \mathrm{T} \ln \left(\frac{2 \mathrm{P}}{\mathrm{P}}\right)=2 \mathrm{RT} \ln 2$
$\Delta \mathrm{W}_{\mathrm{CD}}=1 \times \mathrm{R} \times(\mathrm{T}-2 \mathrm{T})=-\mathrm{RT}$
$\Delta W_{D A}=1 \times R \times T \ln \left(\frac{P}{2 P}\right)=R T \ln \left(\frac{1}{2}\right)$
Net work done in the complete cycle is, $\Delta \mathrm{W}=\Delta \mathrm{W}_{\mathrm{AB}}+\Delta \mathrm{W}_{\mathrm{BC}}+\Delta \mathrm{W}_{\mathrm{CD}}+\Delta \mathrm{W}_{\mathrm{DA}}$
$=\mathrm{RT}+2 \mathrm{RT} \ln 2-\mathrm{RT}+\mathrm{RT} \ln \left(\frac{1}{2}\right)$
$=2 \mathrm{RT} \ln 2+\mathrm{RT} \ln 1-\mathrm{RT} \ln 2$
$=2 \mathrm{RT} \ln 2-\mathrm{RT} \ln 2$
$(\because \ln 1=0)$
$=\mathrm{RT} \ln 2$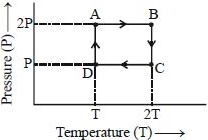
Download our appand get started for free
Similar Questions
- 1View SolutionWhich of the following statements is correct for any thermodynamic system
- 2The temperature of reservoir of Carnot's engine operating with an efficiency of $70\%$ is $1000K.$ The temperature of its sink is ...... $K$View Solution
- 3View SolutionTwo gases are said to be in thermal equilibrium when they have same
- 4View SolutionIn an isothermal change, an ideal gas obeys
- 5An ideal gas follows a process described by the equation $PV ^2= C$ from the initial $\left( P _1, V _1, T _1\right)$ to final $\left(P_2, V_2, T_2\right)$ thermodynamics states, where $C$ is a constant. ThenView Solution
- 6$110\; J$ of heat is added to a gaseous system, whose internal energy change is $40\; J$, then the amount of external work done is ........ $J$View Solution
- 7One mole of an ideal monoatomic gas undergoes the following four reversible processes:View Solution
Step $1$ It is first compressed adiabatically from volume $8.0 \,m ^{3}$ to $1.0 \,m ^{3}$.
Step $2$ Then expanded isothermally at temperature $T_{1}$ to volume $10.0 \,m ^{3}$.
Step $3$ Then expanded adiabatically to volume $80.0 \,m ^{3}$.
Step $4$ Then compressed isothermally at temperature $T_{2}$ to volume $8.0 \,m ^{3}$.
Then, $T_{1} / T_{2}$ is
- 8An ideal gas is taken through the cycle $A \to B \to C \to A,$ as shown in the figure. What is the change in internal energy ...... $J$.View Solution
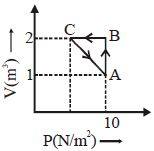
- 9View SolutionWhich statement is incorrect?
- 10View SolutionFirst law of thermnodynamics is given by
