A certain mass of gas at $273 K$ is expanded to $81$ times its volume under adiabatic condition. If $\gamma = 1.25$ for the gas, then its final temperature is ..... $^oC$
Medium
(b) For adiabatic process $T{V^{\gamma - 1}}$= constant
==> $\frac{{{T_2}}}{{{T_1}}} = {\left( {\frac{{{V_1}}}{{{V_2}}}} \right)^{\gamma - 1}}$==> ${T_2} = {\left( {\frac{{{V_1}}}{{{V_2}}}} \right)^{\gamma - 1}} \times {T_1}$
==> ${T_2} = {\left( {\frac{1}{{81}}} \right)^{1.25 - 1}} \times 273$$ = {\left( {\frac{1}{{81}}} \right)^{0.25}} \times 273$
$ = \frac{{273}}{3} = 91K = \,-182°C$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$Assertion :$ In an isolated system the entropy increases.View Solution
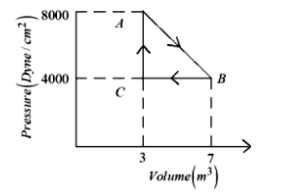
$Reason :$ The processes in an isolated system are adiabatic. - 2View SolutionDuring the thermodynamic process shown in figure for an ideal gas
- 3View SolutionWork done in the cyclic process shown in figure is ...........
- 4A $P-T$ graph is shown for a cyclic process. Select correct statement regarding thisView Solution
- 5An ideal gas expands in such a way that $PV^2 =$ constant throughout the process.View Solution
- 6Work done by a Carnot engine operating between temperatures $127^{\circ}\,C$ and $27^{\circ}\,C$ is $2\,kJ$. The amount of heat transferred to the engine by the reservoir is $........\,kJ$View Solution
- 7Avessel with open mouth contains air at $60^oC$. When the vessel is heated upto temperature $T$, one fourth of the air goes out. The value of $T$ is ..... $^oC$View Solution
- 8Thermodynamic processes are indicated in the following diagram.View Solution
Match the following
$\begin{array}{|l|l|} \hline Column\,\,-\,\,1 & Column\,\,-\,\,2 \\ \hline P\,:\,Process\,\,-\,\,I & \,\,A\,\,:\,\,Adiabatic \\ \hline Q\,:\,Process\,\,-\,\,II & \,\,B\,\,:\,\,Isobaric \\ \hline R\,:\,Process\,\,-\,\,III & \,\,C\,\,:\,\,Isochoric \\ \hline S\,:\,Process\,\,-\,\,IV & \,\,D\,\,:\,\,Isothermal \\ \hline \end{array}$
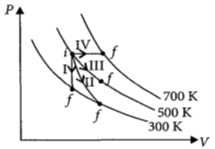
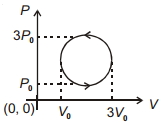
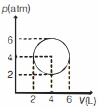
- 9An ideal gas undergoes a circular cycle centred at $4 \,atm , 4 L$ as shown in the diagram. The maximum temperature attained in this process is close toView Solution
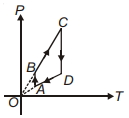
- 10A thermodynamic system is taken from an original state $A$ to an intermediate state $B$ by a linear process as shown in the figure. It's volume is then reduced to the original value from $B$ to $C$ by an isobaric process. The total work done by the gas from $A$ to $B$ and $B$ to $C$ would be :View Solution