The atmospheric pressure at a place is $10^5 \,Pa$. If tribromomethane (specific gravity $=2.9$ ) be employed as the barometric liquid, the barometric height is .......... $m$
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A tank with a small hole at the bottom has been filled with water and kerosene (specific gravity $0.8$). The height of water is $3\,m$ and that of kerosene $2\,m$. When the hole is opened the velocity of fluid coming out from it is nearly ........ $ms^{-1}$ .(take $g\, = 10\, m s^{-2}$ and density of water $= 10^3\, kg\, m^{-3}$)View Solution
- 2View SolutionThe weight of an aeroplane flying in air is balanced by
- 3A log of wood of mass $120 Kg$ floats in water. The weight that can be put on the raft to make it just sink, should be ....... $Kg$ (density of wood = $600 Kg/m^3$)View Solution
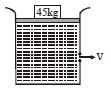
- 4A large cylindrical tank of cross-sectional area $1\ m^2 $ is filled with water. It has a small hole at a height of $1\ m $ from the bottom. $A$ movable piston of mass $5$ $kg$ is fitted on the top of the tank such that it can slide in the tank freely without friction. A load of $45$ $kg$ is applied on the top of water by piston, as shown in figure. The value of $v$ when piston is $7$ $m$ above the bottom is $(g = 10\ m/s^2)$ ....... $m/s$View Solution
- 5View SolutionA wooden piece floats half submerged in a tub of water. If the system is transferred to a lift ascending with acceleration the wooden piece will
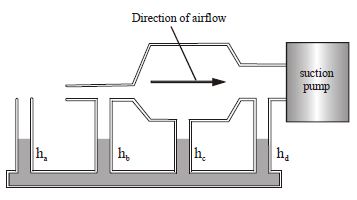
- 6Rank in order, from highest to lowest, the liquid heights $h_a$ to $h_d$ .The air flow is from left to right. The liquid columns are not drawn to scaleView Solution
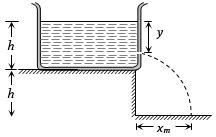
- 7A tank is filled upto a height $h$ with a liquid and is placed on a platform of height h from the ground. To get maximum range ${x_m}$ a small hole is punched at a distance of $y$ from the free surface of the liquid. ThenView Solution
- 8View SolutionA wooden cylinder floats vertically in water with half of its length immersed. The density of wood is
- 9Water is flowing on a horizontal fixed surface, such that its flow velocity varies with $y$ (vertical direction) as $v=k\left(\frac{2 y^2}{a^2}-\frac{y^3}{a^3}\right)$. If coefficient of viscosity for water is $\eta$, what will be shear stress between layers of water at $y=a$.View Solution
- 10A small sphere of mass $m$ is dropped from a great height. After it has fallen $100\; m$ , it has attained its terminal velocity and continues to fall at that speed. The work done by air friction against the sphere during the first $ 100 \;m $ of fall isView Solution
