Two vessels separately contain two ideal gases $A$ and $B$ at the same temperature, the pressure of $A$ being twice that of $B.$ Under such conditions, the density of $A$ is found to be $1.5$ times the density of $B.$ The ratio of molecular weight of $A$ and $B$ is
AIPMT 2015, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A box containing $N$ molecules of a perfect gas at temperature ${T_1}$ and pressure ${P_1}$. The number of molecules in the box is doubled keeping the total kinetic energy of the gas same as before. If the new pressure is ${P_2}$ and temperature ${T_2}$, thenView Solution
- 2View SolutionA tyre kept outside in sunlight bursts off after sometime because of
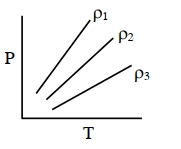
- 3$P-T$ diagram of an ideal gas having three different densities $\rho_1, \rho_2, \rho_3$ (in three different cases) is shown in the figure. Which of the following is correct :View Solution
- 4At which of the following temperature would the molecules of a gas have twice the average kinetic energy they have at $20°C$View Solution
- 5View SolutionThe product of the pressure and volume of an ideal gas is
- 6View SolutionWe sit in the room with windows open. Then,
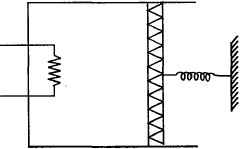
- 7An ideal monoatomic gas is confined in a cylinder by a spring loaded piston of cross section $8.0\times10^{-3}\, m^2$ . Initially the gas is at $300\, K$ and occupies a volume of $2.4\times10^{-3}\, m^3$ and the spring is in its relaxed state as shown in figure. The gas is heated by a small heater until the piston moves out slowly by $0.1\, m$. The force constant of the spring is $8000\, N/m$ and the atmospheric pressure is $1.0\times10^5\, N/m^2$ . The cylinder and the piston are thermally insulated. The piston and the spring are massless and there is no friction between the piston and the cylinder. The final temperature of the gas will be: (Neglect the heat loss through the lead wires of the heater . The heat capacity of the heater coil is also negligible)View Solution
- 8At a given temperature the ratio of $r.m.s.$ velocities of hydrogen molecule and helium atom will beView Solution
- 9The temperature of a gas is $-50^{\circ}\,C$. To what temperature the gas should be heated so that the rms speed is increased by $3$ times?View Solution
- 10A gas is filled in a vessel at a pressure given by $P = \left( {6.02 \times {{10}^{23}}} \right)kT$ where $k$ is the Boltzmann constant and $T$ is the absolute temperature. The number of molecules per unit volume of the gas isView Solution
