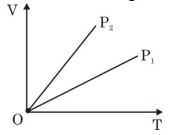
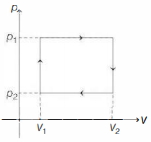
The given figure represents two isobaric processes for the same mass of an ideal gas, then

JEE MAIN 2024, Diffcult
$\mathrm{PV}=\mathrm{nRT}$
$\mathrm{V}=\left(\frac{\mathrm{nR}}{\mathrm{P}}\right)_{\mathrm{T}}$
$\text { Slope }=\frac{\mathrm{nR}}{\mathrm{P}}$
$\text { Slope } \propto \frac{1}{\mathrm{P}}$
$(\text { Slope })_2>(\text { Slope })_1$
$\mathrm{P}_2<\mathrm{P}_1$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionWhich of the following laws of thermodynamics defines the term internal energy?
- 2For an ideal gas, the internal energy is given by $U=5 p V / 2+C$, where $C$ is a constant. The equation of the adiabats in the $p V$-plane will beView Solution
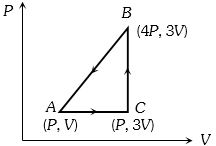
- 3A sample of ideal monoatomic gas is taken round the cycle $ABCA$ as shown in the figure. The work done during the cycle isView Solution
- 4View SolutionAn adiabatic process occurs at constant
- 5The work of $146\ kJ$ is performed in order to compress one kilo mole of gas adiabatically and in this process the temperature of the gas increases by $7^o C$. The gas is $(R=8.3\ J\ mol^{-1} K^{-1})$View Solution
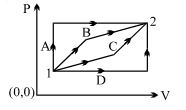
- 6An ideal gas is taken from state $1$ to state $2$ through optional path $A, B, C \& D$ as shown in $P-V$ diagram. Let $Q, W$ and $U$ represent the heat supplied, work done $\&$ internal energy of the gas respectively. ThenView Solution
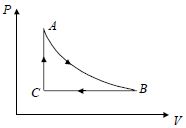
- 7A fixed amount of a gas undergoes a thermodynamic process as shown such that heat interaction along path $B \to C \to A$ is equal to the work done by the gas along path $A \to B \to C$. Then process $A \to B$ is :-View Solution
- 8$n$ moles of a van der Waals' gas obeying the equation of state $\left(p+\frac{n^2 a}{V^2}\right)(V-n b)=n R T$, where $a$ and $b$ are gas dependent constants, is made to undergo a cyclic process that is depicted by a rectangle in the $p-V$ diagram as shown below. What is the heat absorbed by the gas in one cycle?View Solution
- 9View SolutionThe state of an ideal gas was changed isobarically. The graph depicts three such isobaric lines. Which of the following is true about the pressures of the gas?
- 10One mole of an ideal monoatomic gas is heated at a constant pressure of one atmosphere from ${0^o}C$ to ${100^o}C$. Then the change in the internal energy isView Solution
