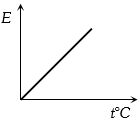
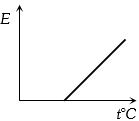
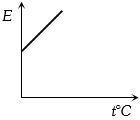
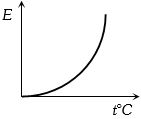
The graph which represent the variation of mean kinetic energy of molecules with temperature $t°C$ is
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A rigid tank contains $35 \,\,kg$ of nitrogen at $6$ atm. Sufficient quantity of oxygen is supplied to increase the pressure to $9$ atm, while the temperature remains constant. Amount of oxygen supplied to the tank is .... $kg$View Solution
- 2View SolutionWhen air is filled in the balloon, the pressure and volume both increases while temperature does not change. Here Boyle's law is not obeyed because
- 3A sample of gas is at $0°C.$ To what temperature it must be raised in order to double the $r.m.s.$ speed of the molecule ....... $^oC$View Solution
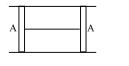
- 4A cylindrical tube of cross-sectional area $A$ has two air tight frictionless pistons at its two ends. The pistons are tied with a straight two ends. The pistons are tied with a straight piece of metallic wire. The tube contains a gas at atmospheric pressure $P_0$ and temperature $T_0$. If temperature of the gas is doubled then the tension in the wire isView Solution
- 5View SolutionThe r.m.s. speed of gas molecules is given by
- 6A spherical bubble inside water has radius $R$. Take the pressure inside the bubble and the water pressure to be $p_0$. The bubble now gets compressed radially in an adiabatic manner so that its radius becomes $(R-a)$. For $a \ll R$ the magnitude of the work done in the process is given by $\left(4 \pi p_0 R a^2\right) X$, where $X$ is a constant and $\gamma=C_p / C_V=41 / 30$. The value of $X$ is. . . . . .View Solution
- 7Relation between $U, P$ and $V$ for ideal gas is $U= 2+2PV$ then gas isView Solution
- 8A polyatomic ideal gas has $24$ vibrational modes. What is the value of $\gamma$ ?View Solution
- 9The molecules of a given mass of a gas have a $r.m.s.$ velocity of $200\, m/sec$ at $27°C$ and $1.0 \times {10^5}\,N/{m^2}$ pressure. When the temperature is $127°C$ and pressure is $0.5 \times {10^5}\,N/{m^2}$, the $r.m.s.$ velocity in $m/sec$ will beView Solution
- 10What is the value of $\frac{R}{{{C_P}}}$ for diatomic gasView Solution